Quantum Dot Antibody Conjugation Service
Bright, Photostable Multiplex ReadoutsBuilt for Imaging, Flow Cytometry, Blotting, and Biosensor Development
Quantum dot antibody conjugation combines the molecular specificity of antibodies with the strong brightness, narrow emission profiles, and high photostability of semiconductor nanocrystal labels. It is especially useful when research teams need direct probes that hold signal during repeated imaging, support cleaner multiplex separation, or expand fluorescence panel design beyond what is practical with conventional dye labels.
We support custom conjugation of whole antibodies, antibody fragments, and secondary detection formats to surface-functionalized quantum dots using application-matched chemistries such as carboxyl-to-amine coupling, thiol-directed linkage, streptavidin-biotin assembly, and oriented click-based strategies where appropriate. Each project is designed around the supplied particle surface, antibody format, target accessibility, excitation/emission requirements, and downstream assay conditions so the final conjugate is aligned with binding performance, colloidal stability, and readout quality.
Why Use Quantum Dot Antibody Conjugation?
Quantum dot antibody conjugation is often chosen when conventional fluorescent probes become the limiting step in an assay. In multiplex staining, overlapping emission channels can make panel design difficult. In long imaging sessions, photobleaching can erode signal confidence. In direct-detection workflows, poor label brightness can make low-abundance targets difficult to resolve. Quantum dots address these problems by providing bright, photostable probes with narrow, well-separated emission channels, while the antibody supplies target specificity. The practical challenge is not simply attaching the antibody to the particle, but doing so in a way that preserves antigen recognition, controls particle behavior, and fits the intended instrument and assay format.
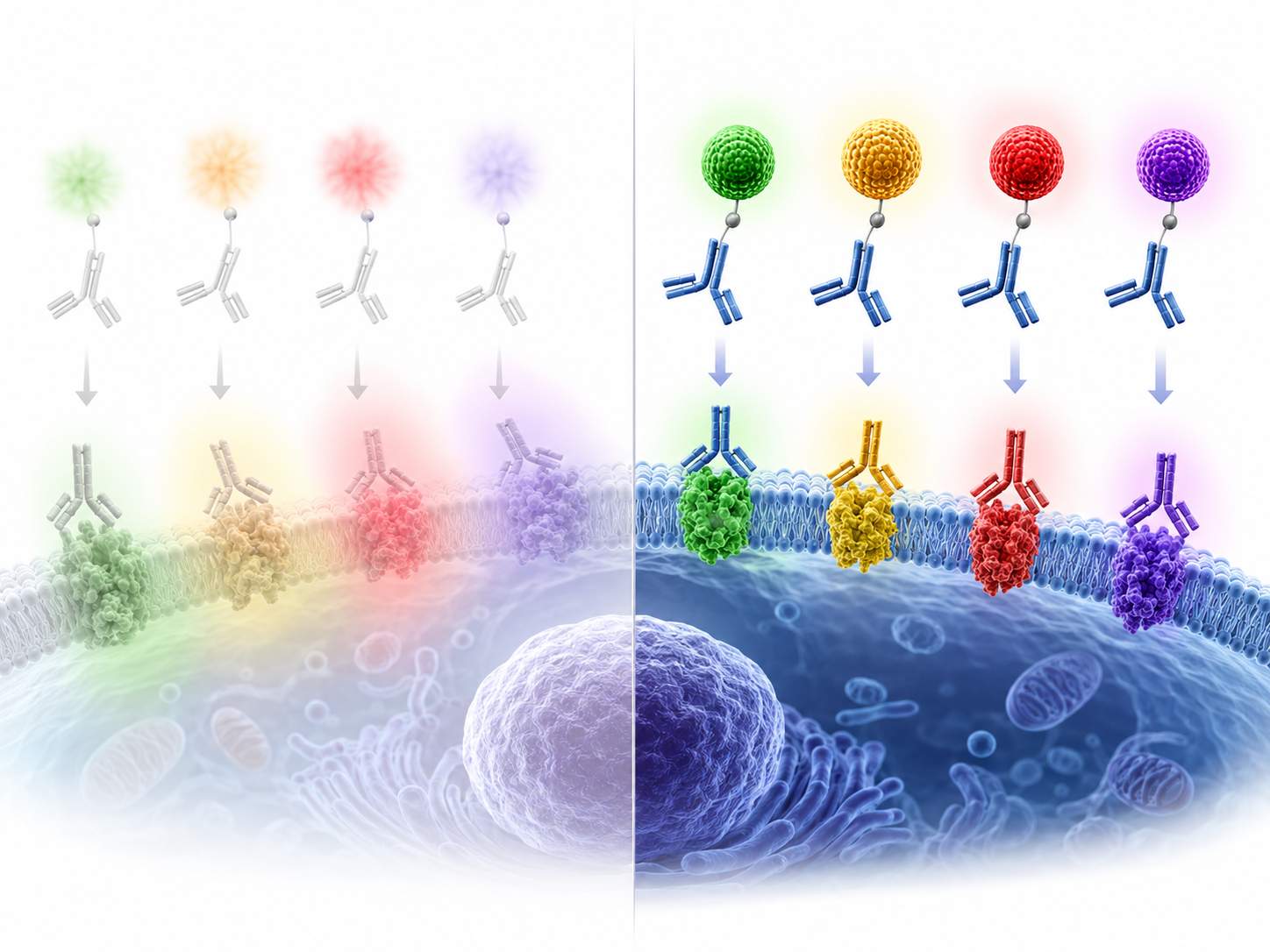 Schematic illustration of quantum dot–antibody probes enabling bright, photostable, multiplex target detection with cleaner spectral separation than conventional dye-only labeling.
Schematic illustration of quantum dot–antibody probes enabling bright, photostable, multiplex target detection with cleaner spectral separation than conventional dye-only labeling.Common Bottlenecks in Quantum Dot Antibody Conjugation Projects
Standard lysine-directed coupling can produce heterogeneous attachment patterns that partially block antigen-binding regions or leave binding domains poorly presented on the particle surface. We help select coupling routes and linker layouts that reduce this risk and improve functional presentation of the antibody on the quantum dot.
A bright quantum dot is not useful if the final conjugate aggregates, adsorbs nonspecifically, or loses colloidal stability in the working buffer. Surface chemistry, purification strategy, and storage conditions are reviewed together to reduce clumping, improve dispersibility, and limit background caused by free antibody or poorly conditioned particles.
Full IgG plus a quantum dot can create a relatively large probe, which may reduce access to crowded epitopes, intracellular structures, or tightly packed assay surfaces. For these cases, we can evaluate fragment-based or more orientation-controlled formats to improve accessibility without giving up the optical advantages of the quantum dot.
Successful quantum dot antibody conjugation requires more than a color change or fluorescence readout. Teams often need evidence that the antibody remains active, free label has been reduced, the particle size remains acceptable, and the final conjugate performs under the intended assay conditions. Our workflows combine physical characterization with binding-focused verification to support confident downstream use.
Our Quantum Dot Antibody Conjugation Services
We provide custom quantum dot antibody conjugation support for research and assay-development teams working in imaging, flow cytometry, blot-based detection, and biosensor platforms. Projects can be configured around client-supplied antibodies, antibody fragments, or quantum dots, with chemistry and characterization selected according to the final application rather than a one-method-fits-all labeling workflow.
 Whole Antibody Quantum Dot Conjugation
Whole Antibody Quantum Dot Conjugation
Capabilities include:
- Conjugation of full-length IgG and related antibody formats to carboxyl-, amine-, thiol-, or affinity-functional quantum dots
- Selection of covalent or affinity-based coupling routes according to supplied surface chemistry
- Buffer review to address azide, carrier proteins, glycerol, or incompatible amines before labeling
- Emission-channel selection aligned with multiplex imaging or cytometry requirements
- Pilot-scale conjugation for feasibility assessment before larger batch preparation
- Removal of unconjugated antibody or excess reactive components as needed
- Preparation of direct-detection probes for antibody-based readouts
- Reporting focused on practical assay deployment
Typical applications:
Direct immunofluorescence, cell and tissue staining, multicolor flow cytometry, membrane-based detection, and antibody-functionalized assay probes
 Oriented and Site-Selective Conjugation Strategy Design
Oriented and Site-Selective Conjugation Strategy Design
Capabilities include:
- Evaluation of Fc-directed, thiol-directed, or click-oriented approaches for improved antibody presentation
- Comparison of random amine coupling versus more controlled labeling strategies
- Linker and spacer selection to balance accessibility, stability, and signal performance
- Controlled reduction planning where sulfhydryl-based attachment is preferred
- Assessment of whether intact antibody or smaller formats are better suited to the target
- Strategy selection based on downstream imaging, cytometry, or sensor constraints
- Minimization of binding-site disruption during conjugation
- Feasibility recommendations before full project execution
Typical applications:
Low-abundance antigen detection, multiplex panel development, receptor-targeting studies, and projects where probe accessibility or functional orientation is critical
 Antibody Fragment and Small-Format QD Conjugates
Antibody Fragment and Small-Format QD Conjugates
Capabilities include:
- Support for Fab, F(ab')2, scFv, VHH, or other client-supplied small antibody formats when lower steric burden is required
- Conjugation planning for targets that are difficult to reach with larger IgG-based probes
- Review of fragment stability and reactive group availability before coupling
- Surface loading control to reduce crowding on the quantum dot
- Optimization for dense epitopes, intracellular targets, or structurally restricted assay formats
- Comparative format guidance when full antibody performance is suboptimal
Focus areas:
Compact probe design, improved target accessibility, and better fit for multiplex imaging or advanced analytical workflows
 Purification, Characterization, and Assay-Fit Optimization
Purification, Characterization, and Assay-Fit Optimization
Capabilities include:
- UV-Vis and fluorescence review of labeled particle signal profiles
- Size and dispersion assessment by DLS and related particle-stability methods
- Zeta potential or equivalent surface-behavior evaluation where relevant
- Gel- or chromatography-based assessment of free antibody and conjugate distribution
- Binding verification using application-matched immunoassay or cell-based methods
- Buffer optimization for storage and short-term working use
- Comparison of pilot lots for consistency review
- Technical reporting focused on practical next-step decision making
Deliverables:
Characterization summary, conjugation observations, particle-behavior data, and assay-relevant recommendations for downstream use
Key Technical Variables That Determine QD–Antibody Conjugate Performance
Strong signal alone does not guarantee a useful quantum dot antibody conjugate. Performance depends on how particle chemistry, antibody format, conjugation route, and downstream assay conditions work together. The table below highlights the main variables that usually need to be aligned early in project planning.
| Design Parameter | Common Options | Why It Matters | Typical Trade-Off | Project Impact |
| Quantum Dot Surface Chemistry | Carboxyl, amine, thiol, maleimide, streptavidin, azide/click-ready surfaces | Determines which conjugation reactions are practical and how stable the final construct is in buffer | A convenient reactive group may not provide the best antibody orientation or background profile | Directly affects feasibility, purification strategy, and reproducibility |
| Antibody Format | Whole IgG, secondary antibody, Fab, F(ab')2, scFv, VHH | Probe size and target accessibility are strongly influenced by the antibody format used | Smaller formats can improve access but may require different handling and coupling logic | Important for dense epitopes, intracellular structures, and multiplex imaging |
| Conjugation Route | EDC/NHS, thiol-maleimide, streptavidin-biotin, copper-free click, affinity-mediated assembly | Chemistry controls stability, orientation, and the degree of heterogeneity in the final conjugate | Fast routes can be simple to execute but may give less control over functional presentation | A key determinant of binding retention and lot-to-lot consistency |
| Spacer and Linker Design | Short, flexible, or application-specific spacing strategies | The distance between antibody and particle can influence steric accessibility and nonspecific behavior | Excessively short linkers may crowd the antibody; excessively long ones can reduce control | Impacts target engagement, background, and assay robustness |
| Emission Channel Selection | Visible to near-IR quantum dot channels selected around instrument filters and panel architecture | The optical channel must fit the intended multiplex design and available excitation sources | A bright color is not useful if it overlaps with existing probes or instrument limits | Critical for flow panels, multicolor imaging, and multiplex membrane assays |
| Purification and Storage Conditions | Spin filtration, size-based cleanup, chromatography, optimized working or storage buffer | Free antibody, excess reactants, and unstable buffers can increase background or reduce shelf performance | More aggressive cleanup can improve purity but may lower recovery | Shapes practical usability and assay transfer readiness |
Conjugation Routes and When to Use Them
There is no single best quantum dot antibody conjugation method for every project. The right route depends on the quantum dot surface, the antibody format, how much control is needed over orientation, and whether the final conjugate will be used for screening, multiplex imaging, cytometry, or assay development.
| Conjugation Strategy | Typical Reactive Pairing | Best Fit | Main Advantages |
| EDC/NHS Coupling | Carboxyl-functional quantum dots to antibody amines | General-purpose covalent conjugation when carboxylated particles are available | Widely used, straightforward to implement, and compatible with many supplied particle formats |
| Thiol-Maleimide Conjugation | Maleimide-activated quantum dots to sulfhydryl groups on reduced antibodies or fragments | Projects that need more controlled attachment than random amine coupling | Strong covalent linkage with useful control when antibody reduction is carefully managed |
| Streptavidin-Biotin Assembly | Streptavidin quantum dots with biotinylated antibodies | Rapid screening, modular probe building, and flexible indirect formats | Fast assembly, high affinity, and simple interchange of different antibodies on a common quantum dot platform |
| Fc-Glycan-Directed Click Labeling | Intact IgG carbohydrate-domain modification followed by copper-free click conjugation | Whole-antibody projects where preserving the antigen-binding region is a priority | More orientation-aware labeling that can help maintain binding-site accessibility |
| Fragment-Based Conjugation | Quantum dots coupled to Fab, scFv, VHH, or other reduced-size binding formats | Restricted targets, dense epitopes, and workflows sensitive to steric hindrance | Lower probe bulk and improved access in applications where full IgG formats are too large |
Analytical Characterization and QC Framework for Quantum Dot–Antibody Conjugates
Useful quantum dot antibody conjugates are defined by both optical signal and biological function. Our characterization framework is designed to confirm that the particle remains fluorescent, the conjugate stays physically stable, the antibody still binds appropriately, and the final material is suitable for the intended assay environment.
| Analytical Category | Methodology | Purpose in Development | Data Delivered |
| Optical Characterization | UV-Vis absorbance and fluorescence spectroscopy | Confirms retained quantum dot signal behavior and checks whether emission remains suitable for the intended readout | Absorbance and emission profile summary |
| Size and Colloidal Stability | Dynamic light scattering and related dispersion measurements | Evaluates hydrodynamic size shift after conjugation and identifies aggregation risk | Size distribution and stability observations |
| Surface Behavior Assessment | Zeta potential or equivalent surface-charge evaluation | Helps interpret suspension stability and interaction tendencies in working buffers | Surface-behavior summary for formulation decisions |
| Conjugate Distribution and Cleanup Review | Gel-based assessment, filtration profile, or size-based separation methods as appropriate | Determines whether free antibody, free particles, or overly broad conjugate populations remain | Purification and conjugate-distribution summary |
| Binding Activity Verification | ELISA-style binding checks, cell-based staining, flow cytometry, or target-specific functional assays | Confirms that labeling has not eliminated target recognition | Application-matched binding performance data |
| Stability Review | Short-term storage assessment under defined handling and light-protection conditions | Supports practical handling recommendations for downstream experimental use | Working-use and storage observations |
Project Workflow for Custom Quantum Dot Antibody Conjugation

We start by reviewing the antibody type, target class, assay goal, supplied quantum dot surface chemistry, desired emission channel, and known buffer constraints. This helps identify feasibility risks early, such as incompatible storage additives, insufficient antibody concentration, or a particle format that does not match the intended readout.
Based on the project requirements, we select the most suitable coupling logic, whether covalent, affinity-based, fragment-oriented, or site-selective. Small-scale pilot work can be used to compare routes when orientation, background, or accessibility are likely to influence final performance.
The conjugation step is executed under conditions chosen to protect antibody function while maintaining particle integrity. Where necessary, linker spacing, reduction level, or attachment density is adjusted to reduce crowding and preserve usable target binding.
Excess reactants, free antibody, or unstable formulation components are addressed through application-appropriate cleanup and buffer conditioning. This stage is important for reducing background, improving colloidal behavior, and preparing the conjugate for real assay use rather than only proof-of-labeling measurements.
We characterize the final conjugate using optical and particle-focused methods, then verify target recognition using the most relevant assay format available for the project. For multiplex workflows, channel fit and signal behavior are reviewed against the planned instrument setup.
Final reporting is structured to help the project team decide how to deploy, scale, or refine the conjugate. Recommendations may include preferred buffers, format changes, additional purification needs, or whether an alternate antibody format would better match the downstream application.
Why Teams Choose Our Quantum Dot Antibody Conjugation Support
We choose the conjugation route based on how the final material will actually be used, not simply on which reactive group is easiest to couple. This helps align probe design with imaging, cytometry, blotting, or biosensor performance goals.

Some targets perform well with full IgG, while others benefit from Fab, scFv, or VHH-style formats. We help match the binding format to the particle and the biological access challenge rather than forcing every project into a full-antibody workflow.
Brightness is only one part of a usable conjugate. We pay close attention to antibody presentation, colloidal stability, cleanup, and working-buffer behavior so the final probe is more likely to perform in real assay conditions.
Our output is designed to help teams move forward with confidence by linking physical characterization to functional performance, so project decisions are based on more than appearance or raw fluorescence alone.
Common Research and Assay Applications for Quantum Dot–Antibody Conjugates
Multiplex Immunofluorescence and Cell/Tissue Staining
- Direct staining strategies where bright, photostable probes improve repeated imaging confidence.
- Multicolor experiments that benefit from narrow emission channels and lower spectral crosstalk.
- Fixed-cell or tissue workflows requiring stronger signal persistence than conventional dye-only labels.
Multicolor Flow Cytometry
- Panel expansion using quantum dot channels excited by common UV or violet-capable instruments.
- Direct or indirect antibody probe formats for improved multicolor flexibility.
- Projects where panel architecture and channel separation are just as important as absolute brightness.
Western Blot and Membrane-Based Multiplex Detection
- Fluorescent blot readouts where longer-wavelength quantum dot probes can support lower-background signal collection.
- Multiplex membrane assays that need cleaner separation of several targets on one blot.
- Workflows seeking direct quantitative fluorescence rather than repeated strip-and-reprobe cycles.
Biosensors and Analytical Assay Development
- Antibody-functionalized fluorescent particles for signal-generating sensor formats.
- Assay-development projects where particle brightness and antibody specificity must be combined in one probe.
- Custom analytical systems requiring tailored particle surface chemistry and antibody presentation.
Discuss Your Quantum Dot Antibody Conjugation Project with Our Scientific Team
Whether you are building a direct quantum dot primary antibody probe, comparing oriented versus random conjugation routes, or troubleshooting aggregation and background in an existing QD-antibody system, we can help structure the project around the final assay requirement.
We work with research teams to evaluate surface chemistry, antibody format, optical channel selection, purification needs, and analytical verification so the resulting conjugate is more practical to deploy in imaging, flow cytometry, blotting, or biosensor workflows.Contact our scientific team to discuss your quantum dot antibody conjugation strategy and request a project-specific feasibility review.
Frequently Asked Questions (FAQ)
It is the process of attaching an antibody or antibody fragment to a quantum dot so the particle provides fluorescence while the antibody provides target specificity.
Whole IgG, secondary antibodies, and smaller formats such as Fab, scFv, or VHH can all be considered, depending on the target-access and assay requirements.
Site-selective approaches are usually preferred when antibody orientation, binding-site accessibility, or conjugate consistency is especially important.
Smaller formats can reduce steric hindrance and improve access to dense or structurally restricted targets where a full IgG–QD probe may be too bulky.
Yes. Quantum dots are often chosen for multiplex workflows because their emission peaks are relatively narrow and their signals are highly photostable.

