GalNAc-Conjugated Oligonucleotide
High-Affinity Hepatocyte TargetingNext-generation Oligonucleotide DeliveryASGPR-Mediated Liver Delivery for siRNA & ASO Programs
Advance liver-targeted nucleic acid therapeutics with GalNAc-conjugated oligonucleotides designed for enterprise R&D and development teams in biotech, pharmaceuticals, and CRO/CDMO settings. GalNAc (N-acetylgalactosamine) conjugation leverages high-affinity binding to the asialoglycoprotein receptor (ASGPR) on hepatocytes, enabling efficient uptake of siRNA and antisense oligonucleotides (ASOs) and supporting clinically validated, subcutaneous dosing strategies used across modern RNA therapeutics pipelines.
We support custom GalNAc–oligonucleotide conjugation for diverse modalities—including siRNA, ASO, and other oligo formats—by integrating conjugation design with sequence, chemistry, and formulation needs typical of discovery-to-IND workflows. Projects can be tailored by GalNAc architecture (commonly triantennary formats), linkage strategy, and oligonucleotide chemistry (e.g., phosphorothioate backbones and 2'-modifications where appropriate), with analytical characterization aligned to development and regulatory expectations.
What Are GalNAc-Conjugated Oligonucleotides?
GalNAc-conjugated oligonucleotides are liver-targeted nucleic acids in which a GalNAc ligand is covalently attached to an oligonucleotide (such as siRNA or an antisense oligo) to promote hepatocyte uptake through ASGPR binding. This receptor-mediated endocytosis approach has become a widely adopted strategy in RNA therapeutics because it can increase hepatic delivery efficiency and improve pharmacologic activity in the liver relative to unconjugated oligonucleotides. In practical development terms, GalNAc conjugation is commonly paired with established oligonucleotide stabilization chemistries and is used to support systemic administration (often subcutaneous) for liver-directed gene silencing or splice modulation programs.
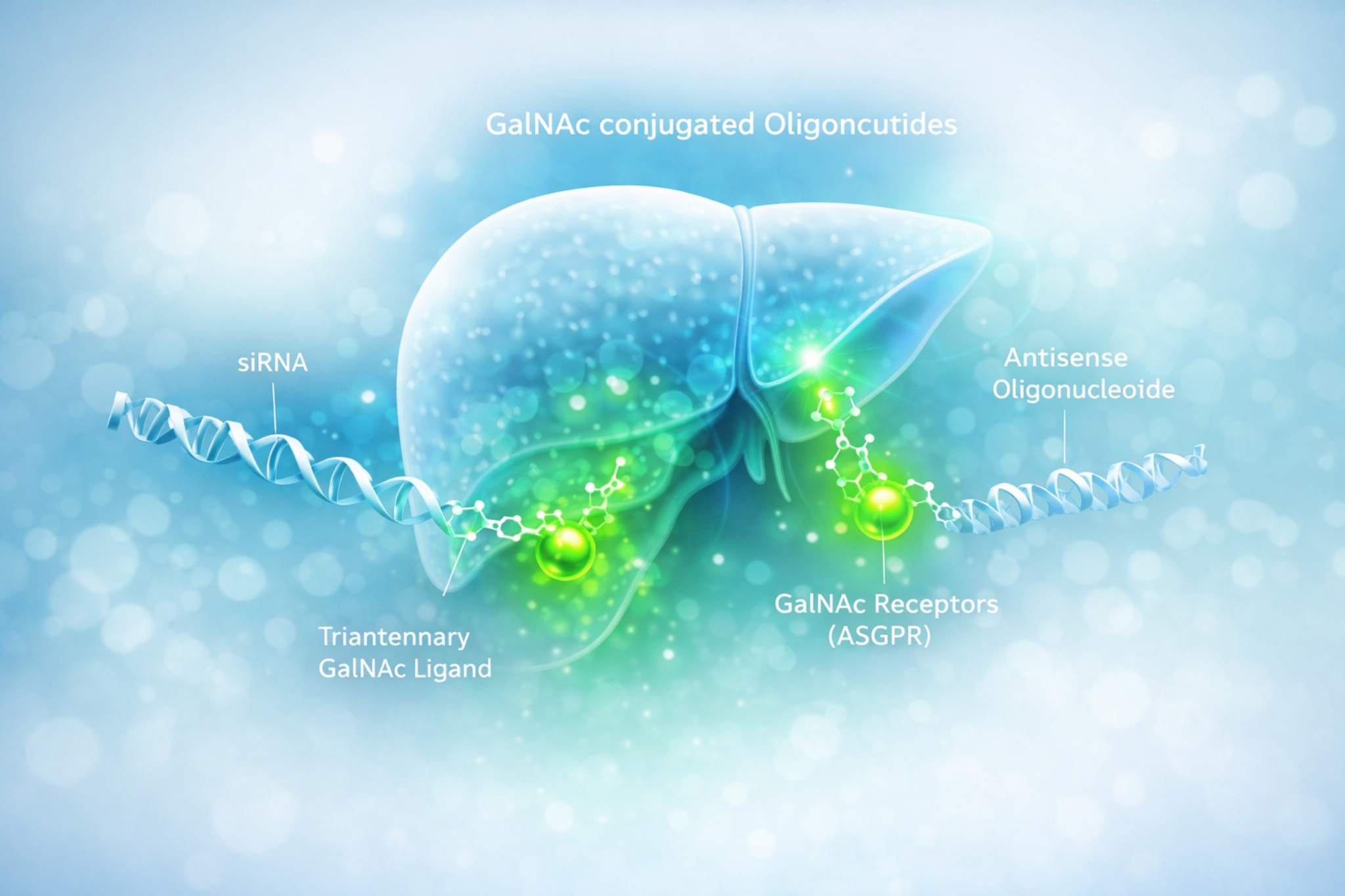 Schematic depiction of triantennary GalNAc–oligonucleotide conjugates enabling ASGPR-mediated hepatocyte uptake for liver-targeted RNA therapeutics.
Schematic depiction of triantennary GalNAc–oligonucleotide conjugates enabling ASGPR-mediated hepatocyte uptake for liver-targeted RNA therapeutics.Key Challenges in Developing GalNAc-Conjugated Oligonucleotide Therapeutics
Suboptimal GalNAc valency, linker design, or conjugation position can reduce binding affinity to ASGPR and limit hepatocyte uptake. We support rational GalNAc architecture selection—commonly triantennary configurations—and controlled conjugation strategies to help maintain high receptor engagement and reproducible liver delivery performance in preclinical programs.
Improper conjugation chemistry or steric hindrance may interfere with RISC loading (for siRNA) or RNase H activity (for ASO). We design conjugation sites and linkers to preserve gene-silencing or splice-modulating activity while enabling efficient hepatic uptake.
Transitioning from discovery-scale synthesis to larger preclinical batches often introduces variability in purity, conjugation efficiency, and analytical profiles. Our workflows emphasize scalable solid-phase synthesis compatibility, controlled conjugation conditions, and batch-to-batch consistency aligned with enterprise development requirements.
IND-enabling programs require comprehensive analytical data, including identity, purity, conjugation integrity, and stability profiles. We integrate HPLC, LC-MS, and spectroscopic verification to generate documentation packages suitable for internal review and regulatory interactions.
Our GalNAc-Conjugated Oligonucleotide Development Services
We provide enterprise-focused GalNAc conjugation services for liver-targeted oligonucleotide therapeutics, supporting discovery, preclinical optimization, and early-stage development. Each project integrates ligand design, conjugation chemistry, and analytical validation to align with current RNA therapeutic market standards and regulatory expectations.
 GalNAc-Conjugated siRNA Development
GalNAc-Conjugated siRNA Development
Capabilities include:
- Conjugation of triantennary GalNAc ligands to sense strands or designated positions for ASGPR-mediated hepatocyte uptake
- Compatibility with commonly used siRNA stabilization chemistries, including phosphorothioate linkages and 2'-O-modifications
- Linker strategy selection to balance stability and intracellular release
- Optimization for subcutaneous administration models in liver-targeted indications
- Analytical verification by HPLC and LC-MS for identity and conjugation integrity
- Support for lead optimization and candidate selection workflows
- Batch reproducibility suitable for preclinical studies
- Documentation aligned with enterprise development standards
Typical applications:
Liver-targeted gene silencing, rare genetic liver diseases, cardiometabolic targets, and infectious disease programs involving hepatic pathways
 GalNAc-Conjugated Antisense Oligonucleotides (ASO)
GalNAc-Conjugated Antisense Oligonucleotides (ASO)
Capabilities include:
- Site-controlled GalNAc attachment to antisense oligonucleotides for targeted hepatic delivery
- Compatibility with gapmer designs and phosphorothioate backbones
- Preservation of RNase H-mediated target RNA degradation
- Conjugation strategies tailored for splice modulation or transcript knockdown applications
- HPLC purification and LC-MS confirmation of final conjugates
- Stability-oriented process design to support in vivo evaluation
- Integration into preclinical pharmacology workflows
- Technical consultation for liver-directed therapeutic strategy
Typical applications:
Splicing correction, mRNA degradation, and liver-specific target modulation in metabolic and genetic disease programs
 Custom GalNAc Ligand & Linker Design
Custom GalNAc Ligand & Linker Design
Capabilities include:
- Design of mono-, bi-, or triantennary GalNAc ligand architectures based on project requirements
- Evaluation of linker length and chemistry to optimize pharmacokinetic and stability profiles
- Conjugation compatibility with diverse oligonucleotide sequences and formats
- Analytical characterization of ligand–oligo conjugation efficiency
- Risk assessment for steric interference and bioactivity preservation
- Technical support for early-stage design decision-making
Focus areas:
Liver-selective delivery optimization, ASGPR binding considerations, and enterprise RNA therapeutic pipeline acceleration
 Analytical Characterization & QC Support
Analytical Characterization & QC Support
Capabilities include:
- Purity assessment via HPLC and UPLC
- Molecular weight confirmation by LC-MS
- Conjugation efficiency evaluation
- Stability studies under defined storage conditions
- Documentation packages for internal R&D and regulatory preparation
- Batch comparison reports to support reproducibility analysis
Deliverables:
Chromatograms, mass spectra, purity percentages, and technical reports aligned with therapeutic development standards
Key Design Parameters for GalNAc-Conjugated Oligonucleotide Therapeutics
Successful development of GalNAc-conjugated oligonucleotides requires careful alignment of ligand architecture, oligonucleotide chemistry, conjugation position, and delivery strategy. The table below summarizes critical technical variables commonly evaluated in liver-targeted RNA therapeutic programs, reflecting current industry standards in GalNAc-siRNA and GalNAc-ASO development.
| Design Parameter | Common Options | Development Considerations | Impact on Therapeutic Profile | Enterprise Relevance |
| GalNAc Architecture | Primarily triantennary GalNAc ligand | Optimized for high-affinity binding to ASGPR on hepatocytes | Enhances receptor-mediated endocytosis and liver selectivity | Industry-standard configuration for clinically advanced liver-targeted RNA therapeutics |
| Conjugation Position | 3′-sense strand (siRNA), terminal 5′ or 3′ (ASO) | Must preserve RISC loading (siRNA) or RNase H activity (ASO) | Influences potency, stability, and intracellular processing | Critical parameter during lead optimization and candidate selection |
| Backbone Chemistry | Phosphorothioate (PS) linkages; mixed PS/PO backbones | Balances nuclease resistance with manufacturability | Affects plasma stability, protein binding, and tissue distribution | Standard component of development-stage oligonucleotide therapeutics |
| Ribose Modifications | 2′-O-methyl, 2′-fluoro, and related chemistries | Integrated to enhance stability and reduce immune stimulation | Improves durability of gene silencing and pharmacokinetic behavior | Required for translational and preclinical program progression |
| Linker Strategy | Stable covalent linkers compatible with systemic administration | Designed to withstand circulation while enabling intracellular functionality | Impacts durability, exposure, and safety margin | Optimization variable during PK/PD refinement |
| Route of Administration | Commonly subcutaneous delivery | Aligned with liver-selective uptake via ASGPR | Supports patient-friendly dosing strategies | Key consideration for commercial viability and lifecycle planning |
GalNAc Conjugation Strategies & Process Development Considerations
GalNAc–oligonucleotide conjugation for therapeutic applications requires precise control over chemistry, scalability, and analytical validation. The following table outlines commonly applied conjugation approaches and their development-stage implications within liver-targeted RNA therapeutic programs.
| Conjugation Strategy | Technical Approach | Common Applications | Development Advantages |
| Solid-Phase Incorporation | GalNAc ligand introduced during oligonucleotide synthesis via modified phosphoramidite chemistry | Clinical-stage siRNA and ASO programs requiring uniform conjugation | High positional control and scalability; suitable for development workflows |
| Post-Synthetic Conjugation | GalNAc ligand attached after oligonucleotide synthesis using controlled covalent chemistry | Discovery and rapid screening studies | Flexible approach for early-stage evaluation and conjugation site comparison |
| Terminal-Specific Conjugation | Attachment at defined 5′ or 3′ termini to maintain functional domains | RNAi and antisense therapeutic constructs | Preserves RISC loading (siRNA) or RNase H activity (ASO) |
| Linker-Optimized Conjugation | Controlled linker length and stability tuning for systemic exposure | PK/PD optimization programs | Enables balance between stability, potency, and durability |
| Scalable Process Design | Conjugation workflows compatible with increasing batch sizes and reproducibility control | Preclinical and IND-enabling material production | Reduces transition risk from discovery to translational stages |
| Analytical Validation Integration | HPLC/UPLC purity assessment, LC-MS identity confirmation, and stability monitoring | Development-stage quality documentation | Supports regulatory-facing documentation and internal CMC review |
IND-Aligned Analytical Characterization & Quality Control Framework
Development of GalNAc-conjugated oligonucleotide therapeutics requires analytical rigor beyond research-grade confirmation. Our analytical framework is structured to support discovery-to-preclinical progression and facilitate preparation for IND-enabling documentation. Methods are designed to ensure identity, purity, conjugation integrity, stability monitoring, and process consistency in accordance with expectations typical of oligonucleotide CMC development.
| Analytical Category | Methodology | Purpose in Development | Data Delivered |
| Identity Confirmation | High-resolution LC-MS / MS analysis | Verification of molecular weight and confirmation of GalNAc conjugation integrity | Mass spectra, deconvoluted molecular weight reports |
| Purity & Impurity Profiling | HPLC / UPLC (ion-exchange or reverse-phase as appropriate) | Quantification of full-length product and assessment of truncated sequences, unconjugated oligo, and process-related impurities | Chromatograms, % purity, impurity distribution profiles |
| Conjugation Efficiency | LC-MS with chromatographic separation | Evaluation of GalNAc attachment uniformity and detection of partially conjugated species | Conjugation ratio analysis and analytical summary report |
| Backbone & Modification Verification | LC-MS fragmentation analysis (when applicable) | Confirmation of phosphorothioate linkages and ribose modifications within development-stage constructs | Structural verification data package |
| Stability-Indicating Analysis | Accelerated and controlled-condition storage studies with periodic HPLC/LC-MS evaluation | Monitoring degradation pathways and conjugation stability | Stability trend data, degradation profile summaries |
| Forced Degradation Assessment | Stress testing under defined thermal or chemical conditions | Identification of potential degradation products for method robustness evaluation | Degradation mapping data supporting analytical method suitability |
| Batch Consistency Evaluation | Comparative chromatographic and mass analysis across production lots | Assessment of reproducibility during scale progression | Lot comparison reports with analytical overlays |
| Documentation Support | Structured analytical reporting aligned with CMC documentation practices | Facilitation of internal review and regulatory submission preparation | Comprehensive analytical report including raw data summaries |
Translational Development Roadmap for GalNAc-Conjugated Oligonucleotides

We begin with a structured evaluation of therapeutic modality (siRNA, ASO), backbone chemistry, target biology, and intended dosing strategy. GalNAc architecture, conjugation position, and linker approach are selected to align with ASGPR-mediated hepatocyte delivery while preserving RNAi or RNase H activity. This stage focuses on reducing early technical risk and accelerating informed lead design decisions.
Rapid production of GalNAc-conjugated variants enables comparative evaluation of conjugation sites, linker configurations, and chemistry combinations. Materials are optimized for in vitro hepatocyte studies and early in vivo liver-targeting models, supporting structure–activity relationship (SAR) refinement and candidate narrowing.
As programs advance, conjugation strategies are refined to improve durability, hepatic exposure, and therapeutic index. Linker stability, conjugation uniformity, and backbone compatibility are evaluated with analytical rigor to support translational pharmacology and dose-interval optimization.
Conjugation workflows are aligned with scalable oligonucleotide synthesis routes, emphasizing reproducibility and impurity control. Batch consistency assessments and analytical comparability studies help reduce transition risk from discovery-scale production to larger preclinical material preparation.
Development-stage analytical methods—including identity confirmation, impurity profiling, conjugation efficiency assessment, and stability-indicating studies—are integrated to support internal CMC documentation. This framework facilitates structured data generation aligned with regulatory submission preparation.
We continue to support program evolution through iterative optimization, batch comparability review, and analytical refinement as therapeutic candidates progress toward translational and early clinical planning stages. Our role extends beyond synthesis to long-term development collaboration.
Strategic Advantages of Our GalNAc-Conjugated Oligonucleotide Development Platform
Our platform is purpose-built for hepatocyte-directed RNA therapeutics, incorporating established triantennary GalNAc strategies and conjugation positioning aligned with current clinical-stage practices. We understand the biological and chemical variables that influence ASGPR uptake and liver-selective pharmacology.

Our conjugation workflows are designed to integrate seamlessly with phosphorothioate backbones, 2′-modified ribose chemistries, and clinically relevant stabilization strategies commonly used in siRNA and antisense therapeutic development.
Beyond synthesis, we integrate development-stage analytical frameworks including impurity profiling, conjugation integrity verification, and stability-indicating studies to support progression toward IND-enabling documentation and internal CMC review.
Conjugation processes are structured for reproducibility and scale transition, reducing technical risk as programs move from discovery-scale batches to larger preclinical material preparation.
Therapeutic Applications of GalNAc-Conjugated Oligonucleotides in Liver-Targeted RNA Medicine
Rare Genetic Liver Diseases
- Gene silencing strategies targeting hepatocyte-expressed pathogenic transcripts.
- Reduction of toxic protein accumulation through RNA interference or antisense modulation.
- Programs designed for durable suppression via subcutaneous GalNAc-siRNA delivery.
Cardiometabolic Disorders
- Targeting liver-expressed genes involved in lipid metabolism and cholesterol regulation.
- RNAi-based reduction of circulating risk factors.
- Development of long-acting GalNAc conjugates for chronic dosing paradigms.
Infectious Disease Targets with Hepatic Expression
- Silencing of viral or host transcripts associated with liver-tropic infections.
- Evaluation of GalNAc-mediated delivery in hepatocyte-driven disease models.
- Integration into combination therapeutic strategies.
Liver-Selective mRNA & Transcript Modulation
- RNase H-mediated antisense approaches targeting hepatocyte transcripts.
- Splice-modulating antisense oligonucleotide programs.
- Selective hepatic knockdown with minimized extrahepatic exposure.
What Our Biotech & Pharmaceutical Partners Say

“Their team demonstrated a strong understanding of GalNAc-siRNA development requirements, particularly around conjugation positioning and analytical comparability. The collaboration helped streamline our lead optimization workflow.”
— Director, RNA Therapeutics Development
“We appreciated their ability to align conjugation strategy with our phosphorothioate ASO design and translational goals. The analytical documentation structure was particularly helpful for internal CMC review.”
— CMC Lead, Biotechnology Company
“Their consultative approach extended beyond synthesis. Discussions around scalability and impurity control added meaningful value as our liver-targeted program advanced toward preclinical development.”
— Head of Oligonucleotide Chemistry, Emerging Biopharma
Advance Your GalNAc-Conjugated Oligonucleotide Program with a Development-Focused Partner
Whether you are refining a discovery-stage GalNAc-siRNA construct, optimizing a liver-directed antisense oligonucleotide, or preparing analytical documentation for translational progression, our team provides technically aligned support across the development lifecycle.
We work closely with RNA biology, pharmacology, and CMC teams to design scalable conjugation strategies, ensure analytical robustness, and reduce technical risk as programs advance toward preclinical and IND-enabling stages.
Contact our scientific team to discuss your GalNAc-conjugated oligonucleotide development strategy and explore a collaboration tailored to your therapeutic objectives.
Frequently Asked Questions (FAQ)
GalNAc conjugation refers to the covalent attachment of N-acetylgalactosamine (GalNAc) ligands to an oligonucleotide, typically siRNA or antisense oligonucleotides (ASOs). The GalNAc moiety binds to the asialoglycoprotein receptor (ASGPR), which is highly expressed on hepatocytes. This receptor-mediated uptake enables selective liver targeting and has become a widely adopted strategy for delivering RNA therapeutics via subcutaneous administration.
GalNAc is used because it enables high-affinity binding to ASGPR on hepatocytes, allowing efficient and selective liver uptake without requiring lipid nanoparticle formulations. This approach supports systemic dosing while maintaining tissue specificity, making it particularly suitable for liver-expressed gene targets.
GalNAc conjugation is commonly applied to:
siRNA (RNA interference therapeutics)
Antisense oligonucleotides (RNase H-mediated degradation)
Splice-modulating oligonucleotides
Other liver-directed nucleic acid constructs
Conjugation must be designed to preserve the biological activity of the oligonucleotide while enhancing hepatocyte delivery.
When properly positioned (often on the 3' end of the sense strand), GalNAc conjugation enhances hepatocyte uptake while maintaining RISC loading and gene-silencing activity. Improper conjugation design, however, may interfere with intracellular processing, which is why positional and linker optimization are critical during development.
GalNAc conjugation enables direct, receptor-mediated liver targeting without the need for lipid nanoparticle encapsulation. Compared to LNP systems, GalNAc-conjugated oligonucleotides often support subcutaneous administration and may offer a simplified formulation approach for liver-specific indications. The optimal strategy depends on therapeutic context and target biology.

