Antibody Conjugated LNP
Cell-Selective RNA DeliveryAntibody-Guided Surface EngineeringCustom Ab-LNP Development for Discovery and Preclinical Research
Antibody-conjugated lipid nanoparticles (Ab-LNPs) combine the payload capacity of LNPs with the binding specificity of antibodies, enabling more selective delivery of mRNA, siRNA, ASO, plasmid DNA, or related cargos to defined cell populations. For teams working beyond passive biodistribution alone, Ab-LNP design offers a practical route to improve receptor engagement, enhance cell uptake, and build targeted delivery systems that are better aligned with real biological questions.
We support custom antibody-conjugated LNP development for research, screening, and preclinical programs by integrating antibody selection, linker and spacer design, LNP formulation, conjugation process development, and analytical characterization into one workflow. Depending on project needs, we can support whole antibodies, Fab fragments, or engineered binders; conventional coupling routes such as thiol-reactive or amine-reactive chemistry; and orthogonal surface-engineering options informed by bioorthogonal click chemistry, PEGylation, and related nanoparticle conjugation strategies.
What Problems Can Antibody-Conjugated LNPs Help Solve?
Standard LNPs are powerful delivery vehicles, but many projects reach a point where payload encapsulation alone is not enough. Researchers may obtain acceptable particle size and encapsulation efficiency yet still see weak delivery to the intended cell population, broad off-target uptake, or unclear structure-activity relationships once a targeting ligand is added. Antibody-conjugated LNPs are developed to address these bottlenecks by placing a cell-recognition element on the particle surface, allowing the formulation to engage selected receptors and support a more deliberate active-targeting strategy. In practice, this approach is especially valuable when programs need to improve delivery to difficult-to-transfect cells, compare receptor-specific uptake pathways, reduce wasted screening cycles caused by poorly oriented antibodies, or preserve LNP performance after surface modification. The goal is not simply to add an antibody to an LNP, but to create a conjugated system in which target recognition, particle stability, cargo protection, and downstream assay performance remain aligned.
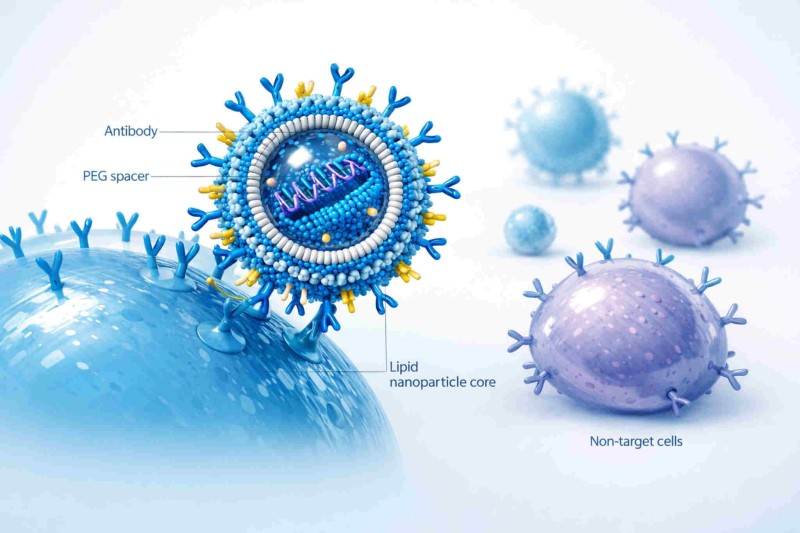 Conceptual schematic of antibody-conjugated LNPs using surface-displayed antibodies to improve receptor-specific binding, internalization, and payload delivery to target cells.
Conceptual schematic of antibody-conjugated LNPs using surface-displayed antibodies to improve receptor-specific binding, internalization, and payload delivery to target cells.
Key Challenges in Antibody-Conjugated LNP Development
Random modification of lysines or uncontrolled thiol exposure can reduce antigen recognition, alter antibody structure, or create poorly oriented surface display. We help evaluate coupling routes, spacer architecture, and antibody format so the final Ab-LNP is built for target engagement rather than simple attachment alone.
Adding antibodies or reactive lipids may shift particle size distribution, increase aggregation risk, or affect payload retention. Our workflows focus on preserving critical formulation attributes while introducing the targeting layer under controlled conditions.
Too little antibody can lead to weak receptor binding, while excessive surface loading may create steric crowding, poor colloidal behavior, or misleading uptake data. We support density screening and structure-function comparison so design decisions are based on measurable performance, not assumptions.
Many projects can confirm particle formation but still lack direct evidence for successful conjugation, antibody integrity, or receptor-binding retention. We combine physicochemical and functional characterization to verify both the LNP and the antibody layer in a project-relevant way.
Our Antibody-Conjugated LNP Development Services
We provide development-focused Ab-LNP support for teams that need more than a generic formulation package. Services can be configured around your target receptor, antibody source, payload type, assay plan, and preferred conjugation route, with optional alignment to related antibody liposome conjugation or broader liposome conjugation programs when comparative delivery systems are required.
 Target and Antibody Feasibility Assessment
Target and Antibody Feasibility Assessment
Capabilities include:
- Review of target receptor biology, internalization behavior, and intended cell population
- Assessment of full antibody, Fab, scFv, or other binder formats for surface display suitability
- Evaluation of available reactive handles on the antibody and compatibility with planned chemistry
- Early-stage risk analysis for orientation, steric hindrance, and assay interference
- Recommendation of screening matrix for linker, density, and formulation variables
Typical use:
Projects that need to determine whether a receptor-targeting concept is technically realistic before larger formulation campaigns begin
 Antibody Surface Conjugation Strategy Development
Antibody Surface Conjugation Strategy Development
Capabilities include:
- Development of covalent coupling routes such as maleimide-thiol, NHS/amine, carbodiimide, or click-enabled approaches
- Selection of reactive PEG-lipids or post-insertion components for antibody presentation
- Spacer and linker design to balance accessibility, stability, and particle behavior
- Comparison of direct conjugation versus post-insertion workflows
- Antibody loading studies to establish workable surface density ranges
Typical use:
Programs optimizing how the antibody is attached, not just whether attachment can be detected
 LNP Formulation and Payload Encapsulation
LNP Formulation and Payload Encapsulation
Capabilities include:
- Formulation of LNPs for mRNA, siRNA, ASO, plasmid DNA, or other nucleic acid cargos
- Screening of ionizable lipid, helper lipid, cholesterol, and PEG-lipid composition
- Adjustment of process variables that affect particle size, PDI, and encapsulation efficiency
- Compatibility assessment between payload-sensitive formulations and downstream conjugation steps
- Comparative development of targeted and non-targeted controls
Typical use:
Teams that need a functional targeted formulation rather than an antibody coupling exercise performed on an unstable particle
 Physicochemical and Functional Characterization
Physicochemical and Functional Characterization
Capabilities include:
- Particle size, PDI, and zeta potential measurement
- Encapsulation efficiency and payload recovery analysis
- Conjugation confirmation by orthogonal analytical methods
- Antibody content, integrity, and surface-display assessment
- Functional binding or cell-association studies for target-specific comparison
Deliverables:
Structured datasets linking surface engineering decisions to measurable Ab-LNP performance
 Optimization for Screening and Preclinical Studies
Optimization for Screening and Preclinical Studies
Capabilities include:
- Side-by-side comparison of antibody clones, densities, and spacer lengths
- Serum-exposure and storage-condition review for formulation robustness
- Targeted versus non-targeted benchmarking in project-relevant assays
- Small-batch iteration cycles to refine candidate designs efficiently
- Support for scale progression once a lead Ab-LNP configuration is selected
Typical use:
Discovery and preclinical teams narrowing multiple Ab-LNP concepts into a smaller, testable lead set
 Custom Documentation and Technical Reporting
Custom Documentation and Technical Reporting
Capabilities include:
- Project summaries covering conjugation route, materials, and critical processing choices
- Analytical result packages with method descriptions and interpreted findings
- Batch comparison summaries for iterative development work
- Troubleshooting notes for unsuccessful or borderline configurations
- Communication support aligned with internal formulation, biology, or CMC teams
Typical use:
Cross-functional programs that need decisions, not just raw data files, at each project milestone
Critical Design Parameters for Antibody-Conjugated LNPs
Successful Ab-LNP development depends on managing both the nanoparticle and the targeting ligand as one integrated system. The table below summarizes design variables that commonly determine whether a targeted formulation remains stable, measurable, and biologically informative.
| Design Parameter | Common Options | Why It Matters | Potential Risk If Poorly Controlled | Project Impact |
| Antibody Format | Whole IgG, Fab, scFv, engineered binder | Determines size, valency, steric profile, and available conjugation handles | Reduced accessibility, aggregation tendency, or low functional display | Directly affects receptor binding behavior and formulation complexity |
| Conjugation Route | Maleimide-thiol, NHS/amine, EDC/NHS, click-enabled ligation, post-insertion | Controls attachment efficiency, selectivity, and processing workflow | Random coupling, low reproducibility, or antibody activity loss | Shapes the balance between speed, control, and manufacturability |
| Spacer / PEG-Lipid Architecture | Reactive PEG-lipids with varied chain length and terminal functionality | Influences antibody accessibility and surface mobility | Steric shielding, poor receptor engagement, or unstable presentation | Often determines whether conjugation translates into functional targeting |
| Antibody Surface Density | Low, moderate, or high loading screens | Affects avidity, colloidal stability, and cell-interaction profile | Weak targeting at low density or crowding and aggregation at high density | A key optimization variable during candidate ranking |
| LNP Composition | Ionizable lipid, helper lipid, cholesterol, PEG-lipid combinations | Governs particle formation, cargo protection, and intracellular behavior | Poor encapsulation, unstable particles, or inconsistent performance after decoration | Sets the baseline performance that targeting chemistry must preserve |
| Payload Type | mRNA, siRNA, ASO, plasmid DNA, other nucleic acid cargos | Different cargos place different demands on formulation and assay design | Mismatched process conditions or misleading functional readouts | Determines encapsulation strategy and downstream validation plan |
| Particle Quality Attributes | Size, PDI, zeta potential, encapsulation efficiency, recovery | Provide a direct readout of formulation integrity after conjugation | Batch drift, poor comparability, or unreliable biological results | Essential for screening confidence and scale progression |
Antibody Attachment Routes and Development Considerations
Different Ab-LNP projects require different coupling logic. Some teams need fast screening on preformed particles, while others prioritize positional control, cleaner analytical interpretation, or easier scale progression. The table below outlines commonly used development routes and when each approach is typically most useful.
| Development Route | Technical Approach | Best Suited For | Main Considerations |
| Direct Covalent Surface Conjugation | Antibody coupled to reactive groups already present on the LNP surface | Programs needing a straightforward initial chemistry route | Requires control over antibody modification extent and surface accessibility |
| PEG-Lipid Post-Insertion | Antibody first linked to a functional PEG-lipid, then inserted into preformed LNPs | Rapid screening, modular target comparison, and flexible binder swapping | Insertion efficiency, ligand retention, and particle stability must be verified |
| Maleimide-Thiol Coupling | Sulfhydryl-reactive conjugation between thiolated antibody regions and maleimide-functional lipids | Projects seeking established chemistry with relatively simple implementation | Thiol generation strategy and over-reduction risk should be controlled carefully |
| Amine-Reactive or Carbodiimide Coupling | Antibody amines react with activated carboxyl-containing surface groups | Early feasibility work when dedicated thiol or click handles are unavailable | More random attachment can complicate orientation and functional consistency |
| Click-Enabled Surface Engineering | Orthogonal ligation using preinstalled click handles on antibody and particle components | Projects prioritizing selective chemistry and cleaner conjugation logic | Handle installation and compatibility with payload-sensitive formulations must be planned in advance |
| Comparative Multi-Route Screening | Several antibody formats or attachment routes evaluated in parallel on a common LNP core | Programs with uncertain target biology or multiple candidate binders | Most informative when analytical and functional endpoints are defined before screening starts |
Analytical Characterization Framework for Ab-LNP Projects
A targeted LNP is only useful when both parts of the system can be verified: the nanoparticle must remain well formed and the antibody layer must still be meaningful. Our characterization framework is built to connect conjugation success with particle quality and functional relevance rather than reporting each dataset in isolation.
| Analytical Category | Representative Methods | What It Confirms | Typical Output |
| Particle Size and Distribution | DLS and related particle-sizing methods | Hydrodynamic diameter and PDI before and after conjugation | Size distribution data and comparability summary |
| Surface Charge Assessment | Zeta potential measurement | Surface-state changes introduced by antibody display or spacer insertion | Charge profile comparison across formulations |
| Encapsulation and Payload Recovery | Fluorescent dye exclusion, nucleic acid quantitation, or project-matched assays | Whether conjugation or processing affects payload retention | Encapsulation efficiency and recovery values |
| Conjugation Confirmation | Orthogonal approaches such as SEC, gel-based analysis, immunoassays, or surface-accessible quantitation | Presence of antibody on the particle and degree of surface modification | Conjugation summary with method-specific evidence |
| Antibody Integrity | SDS-PAGE, CE-SDS, immunoreactivity assays, or other suitable methods | Whether the antibody remains structurally usable after processing | Integrity profile and comparative interpretation |
| Target Binding / Cell Association | ELISA-style binding studies, flow cytometry, microscopy, or cell uptake assays | Retention of target interaction and value of the targeting layer versus controls | Functional comparison between targeted and non-targeted systems |
| Stability and Comparability | Defined storage-condition monitoring and batch comparison studies | Whether the Ab-LNP remains usable over time and across iterations | Stability trend data and lot-to-lot assessment |
Project Workflow for Antibody-Conjugated LNP Development

We begin by defining the target cell population, receptor rationale, payload type, assay needs, and decision criteria for the program. This step ensures the Ab-LNP design is driven by the intended experiment or preclinical objective rather than by chemistry in isolation.
Antibody format, available reactive handles, spacer design, and feasible coupling routes are reviewed together. We define an initial strategy that balances functional display, formulation compatibility, and the level of analytical control needed for the project.
Core LNPs are formulated and then converted into targeted variants through the selected surface-engineering workflow. Non-targeted and process controls are prepared in parallel wherever possible so later performance differences can be interpreted more cleanly.
Particle quality, payload retention, antibody attachment, and antibody integrity are characterized using fit-for-purpose methods. This stage is designed to answer a practical question: did the conjugation create a usable Ab-LNP, or only a measurable modification?
Targeted and non-targeted formulations are compared in project-relevant assays to refine antibody density, linker choice, particle composition, or binder format. The objective is to connect formulation variables with functional outcomes instead of relying on single-parameter optimization.
Once a preferred Ab-LNP configuration is identified, the workflow can move into repeat preparation, batch comparison, and technical reporting to support broader in vitro or in vivo studies. This reduces handoff risk as programs transition from feasibility to more structured development.
Why Teams Choose Our Ab-LNP Development Support
We do not treat antibody coupling as a stand-alone chemistry task. Surface engineering is evaluated alongside particle attributes, cargo retention, and assay readouts so the final design remains usable as an LNP system.

Projects vary widely in antibody format, available handles, and allowable processing conditions. Our development logic adapts to those constraints instead of forcing every program into one coupling route.
We focus on whether the antibody remains useful after conjugation and whether the targeted particle behaves differently from an appropriate control, which is what project teams ultimately need for decision-making.
Ab-LNP programs often advance through comparison rather than one-pass success. We structure work to make failed conditions interpretable and promising conditions easier to reproduce in the next round.
Research Applications of Antibody-Conjugated LNPs
Immune Cell-Targeted RNA Delivery
- Development of Ab-LNPs for selective delivery to defined immune cell subsets.
- Screening of receptor-targeting strategies for mRNA or siRNA payloads.
- Comparison of targeted and non-targeted formulations in cell-association and expression studies.
Endothelial and Stromal Cell Targeting
- Surface engineering of LNPs to evaluate uptake by vascular or stromal cell populations.
- Receptor-driven delivery studies where passive LNP uptake is insufficient.
- Optimization of binder density and spacer effects for difficult-to-access cell types.
Cell-Selective Gene Silencing and Editing Research
- Targeted delivery of siRNA, guide-containing systems, or related nucleic acid cargos.
- Evaluation of receptor-dependent internalization as part of payload performance studies.
- Support for programs that need to separate uptake efficiency from payload potency.
Comparative Targeting-Ligand Screening
- Parallel evaluation of whole antibodies, fragments, or alternative binders on a shared LNP backbone.
- Rapid down-selection of targeting formats before deeper biology studies.
- Useful when receptor choice is defined but ligand architecture is still open.
Biodistribution and Uptake Mechanism Studies
- Design of targeted LNPs for uptake mapping and receptor-dependence experiments.
- Integration with fluorescence or analytical readouts for comparative tracking.
- Suitable for teams trying to explain why an LNP succeeds or fails in a given model.
Platform Development for Next-Generation Delivery Systems
- Support for modular targeted LNP platforms intended for repeated target swapping.
- Optimization of conjugation logic, characterization standards, and lead-selection criteria.
- Useful for groups building reusable targeted delivery workflows rather than single formulations.
Advance Your Antibody-Conjugated LNP Program with a More Structured Development Workflow
Whether you are evaluating a first targeting concept, troubleshooting antibody orientation on a preformed LNP, or narrowing a set of receptor-directed formulations for deeper biological testing, we can help you move from idea to interpretable Ab-LNP data with a workflow built around conjugation, formulation, and characterization together.
Our support is designed for research and preclinical teams that need targeted LNP systems they can compare, optimize, and reproduce—not just particles that appear modified on paper.Contact our scientific team to discuss your antibody-conjugated LNP project, payload type, target receptor, and preferred development pathway.
Frequently Asked Questions (FAQ)
There are multiple conjugation strategies, including:
Carbodiimide reaction: Conjugating carboxyl (-COOH)-containing lipids with primary amines (-NH) on antibodies.
Disulfide bond exchange: Reacting thiol (-SH) groups from cysteine residues in the antibody with pyridyl disulfide reactive groups to form unstable disulfide bonds.
Michael addition: Reacting maleimide groups on PEGylated lipids with thiol groups from partially reduced antibodies.
This approach enhances drug accumulation in tumor tissues, improving anti-tumor efficacy while reducing side effects on normal tissues. The targeting ability of antibodies allows LNP-encapsulated drugs to be precisely delivered to tumor cells, ensuring selective tumor cell destruction.
Strict quality control and characterization are necessary, including:
Physicochemical characterization (e.g., particle size distribution, surface charge, morphology, drug encapsulation efficiency, antibody conjugation efficiency).
Biological activity assays (e.g., antibody immunoactivity, drug release properties, cytotoxicity).
Stability testing to assess Ab-LNP stability under different conditions.
Key indicators include:
Particle size and polydispersity index (PDI): Affecting Ab-LNP stability and delivery efficiency.
Drug encapsulation efficiency and antibody conjugation efficiency: Reflecting drug and antibody loading within LNPs.
Biological activity metrics: Such as cytotoxicity and immunogenicity.
Several factors must be considered, including target specificity, low immunogenicity, conjugation efficiency, particle size, accessibility, and cellular internalization. For instance, the antibody should have high affinity and specificity to accurately recognize and bind to the target antigen. Additionally, antibody size and structure influence the formulation and performance of Ab-LNPs.
Yes, in addition to full-length IgG antibodies, smaller antibody fragments such as Fab and scFv can also be used. These smaller antibodies facilitate deeper penetration into solid tissues or tumors, improving drug delivery efficiency.