Oligonucleotides Conjugated with Small Molecules
Custom Oligo–Ligand DesignFlexible Linker ChemistryPurified and Characterized Conjugates
We provide custom services for oligonucleotides conjugated with small molecules to support delivery research, targeting studies, affinity capture, molecular labeling, and assay development. Our workflow covers oligonucleotide review, conjugation-site planning, small-molecule selection, linker and spacer design, conjugation chemistry, purification, and analytical characterization for DNA, RNA, antisense oligonucleotides, siRNA, aptamers, and other modified oligo formats.
Projects may involve lipophilic modifiers, receptor-targeting ligands, reporter tags, or custom research handles, depending on the intended biological or analytical function. We can support standalone builds or integrate projects with broader oligonucleotide bioconjugation programs, as well as focused needs such as cholesterol conjugation, biotin-labeled oligonucleotides, fluorescence labeling of oligonucleotides, and PEG-conjugated oligonucleotide development.
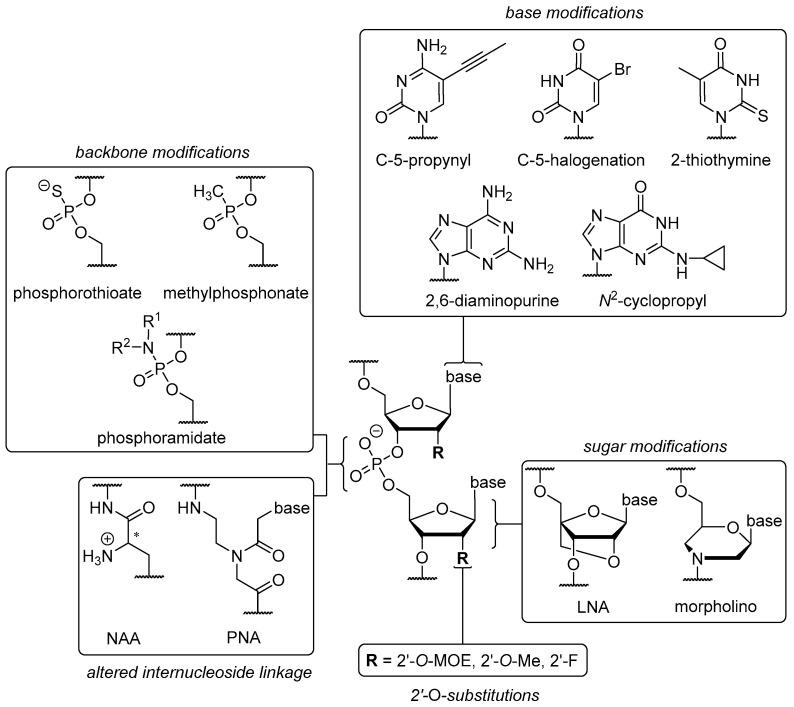 Available structural modification sites of oligonucleotide to small molecule. (Hawner, 2020)
Available structural modification sites of oligonucleotide to small molecule. (Hawner, 2020)
What Problems Can Oligonucleotides Conjugated with Small Molecules Solve?
Many oligonucleotide projects reach a point where sequence optimization alone is not enough. Researchers may have a functional ASO, siRNA, DNA probe, or aptamer, but still face poor cellular uptake, weak tissue selectivity, limited membrane interaction, difficult capture or imaging workflows, or inconsistent behavior after introducing hydrophobic or reporter-type modifications. Oligonucleotides conjugated with small molecules are used to add a defined functional module—such as a delivery modifier, targeting ligand, or affinity label—without redesigning the entire oligo sequence.
In practice, successful conjugate design requires more than simply attaching a ligand at the 5′ or 3′ end. The conjugation site, linker length, oligo chemistry, duplex architecture, hydrophobicity, and purification strategy all influence whether the final construct remains soluble, structurally intact, analytically clean, and biologically useful. This is especially important for projects involving GalNAc, cholesterol, folate, tocopherol, biotin, fluorophores, or custom small molecules where steric effects and free-ligand carryover can directly affect study results.
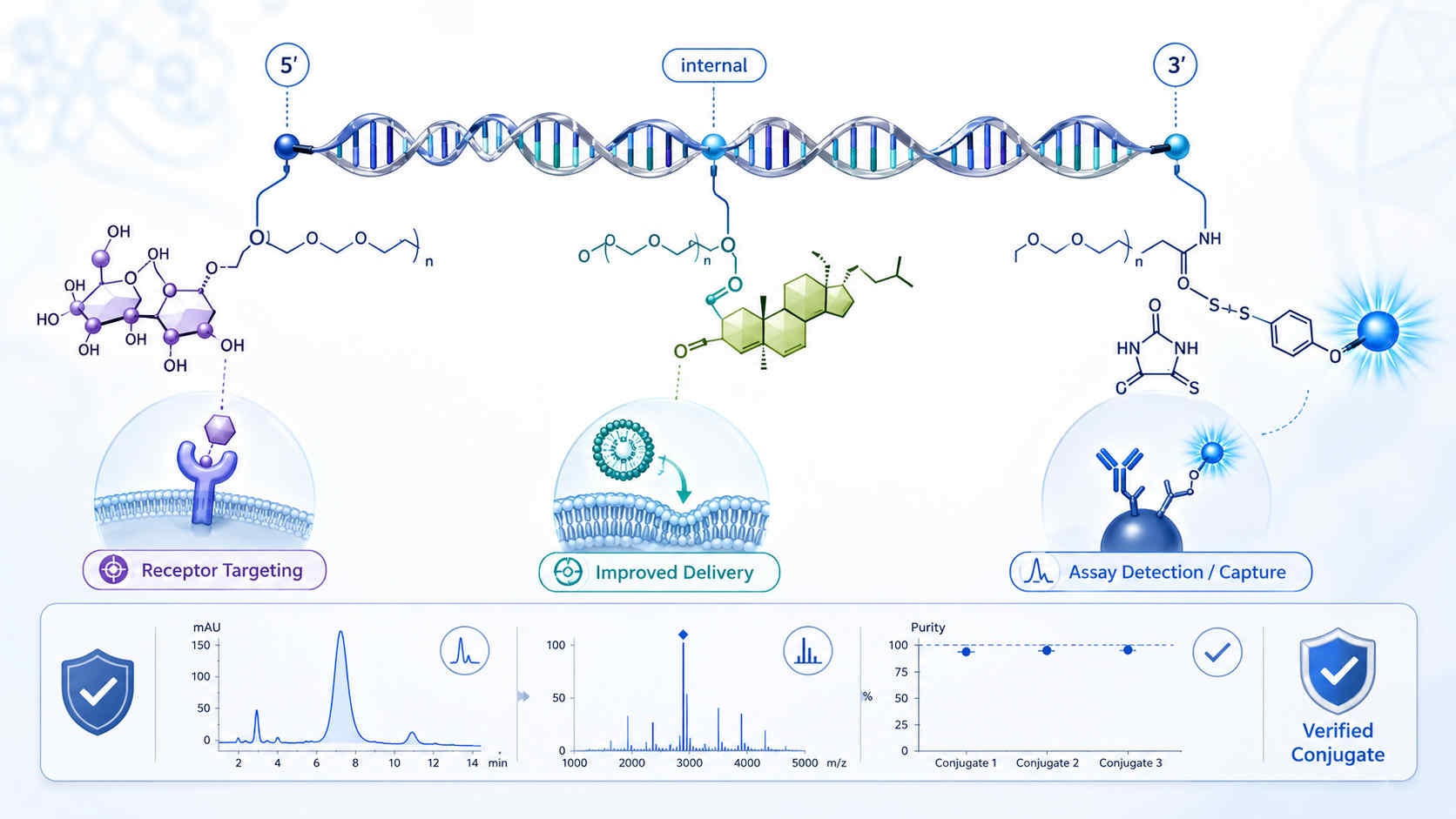 Schematic illustration of oligonucleotide-small molecule conjugation showing how controlled attachment site, linker design, and purification can improve delivery behavior, targeting, and assay performance.
Schematic illustration of oligonucleotide-small molecule conjugation showing how controlled attachment site, linker design, and purification can improve delivery behavior, targeting, and assay performance.Key Challenges Research Teams Face in Oligo–Small Molecule Conjugation
A small molecule that improves uptake can also interfere with duplex formation, target binding, or oligo accessibility if it is placed on the wrong strand, attached too close to the active sequence, or paired with an unsuitable spacer. We help match attachment position and linker design to the functional mechanism of the oligonucleotide rather than treating conjugation as a generic end-labeling step.
Cholesterol, tocopherol, lipophilic dyes, and other hydrophobic moieties can complicate reaction setup, purification, buffer exchange, and storage. Projects often struggle with aggregation, broad chromatographic peaks, and residual free ligand. We design the workflow around the polarity and handling behavior of both the oligo and the attached small molecule.
Many customer-supplied ligands require a practical decision between amino, thiol, azide, alkyne, carboxyl, or phosphate-directed chemistry. Selecting the wrong handle can reduce conversion, create side reactions, or make purification unnecessarily difficult. We review the available reactive groups and choose a chemistry route that balances selectivity, oligo integrity, and manufacturability.
A mass shift alone does not show whether the conjugate is sufficiently pure, whether free ligand has been removed, or whether the modified oligo still behaves correctly in hybridization, uptake, or capture experiments. We combine chemistry-specific purification with orthogonal analytical checks so the final data package supports development decisions instead of only confirming that coupling occurred.
Our Oligonucleotide–Small Molecule Conjugation Services
We provide custom service packages for oligonucleotides conjugated with small molecules, from early feasibility review through purified conjugate delivery and analytical reporting. Projects may start from a customer-defined oligo sequence, an existing modified oligonucleotide, or a customer-supplied ligand that needs to be reformatted for controlled attachment and downstream use.
 Strategy & Handle Design
Strategy & Handle Design
Capabilities include:
- Review of oligonucleotide modality, strand architecture, sequence constraints, and intended function across ASO, siRNA, DNA, RNA, and aptamer formats
- Selection of 5′, 3′, internal, sense-strand, antisense-strand, or post-assembly attachment sites based on activity retention and practical synthesis logic
- Planning of compatible reactive handles such as amino, thiol, azide, alkyne, carboxyl, or phosphate-derived entry points
- Assessment of whether direct conjugation, pre-installed modifier synthesis, or post-synthetic coupling is the better route for the project
- Early identification of structural risks related to steric bulk, hydrophobicity, linker exposure, and duplex stability
Typical applications:
GalNAc-siRNA concepts, cholesterol-modified oligos, ligand-functionalized ASOs, and custom DNA or RNA constructs that require defined site control before conjugation work begins
 Ligand & Label Conjugation
Ligand & Label Conjugation
Capabilities include:
- Conjugation of delivery modifiers, receptor-targeting ligands, reporter tags, and analytical handles to single-stranded or duplex oligonucleotides
- Support for small molecules such as GalNAc-type ligands, cholesterol, tocopherol, folate, biotin, fluorophores, click handles, and other custom research moieties
- Use of stable or application-matched linker architectures depending on whether the project prioritizes uptake, targeting, capture, or signal generation
- Coordination of customer-supplied ligand evaluation, functional-group introduction, or compatibility checks before scale-up
- Optional alignment with broader custom bioconjugation services when the construct requires additional molecular engineering
Typical applications:
Cell uptake studies, receptor-targeted oligonucleotide programs, affinity capture reagents, imaging probe development, and customized research-grade ligand-oligo constructs
 Linker & Process Optimization
Linker & Process Optimization
Capabilities include:
- Spacer design using PEG-like, alkyl, triethylene glycol, or other separation motifs to control sterics, polarity, and ligand presentation
- Chemistry route selection across amide coupling, maleimide-thiol coupling, CuAAC click chemistry, copper-free click chemistry, and EDC/NHS-enabled activation strategies where appropriate
- Adjustment of reaction order, solvent balance, and handle placement to reduce hydrolysis, incomplete conversion, or oligo degradation
- Small matched design sets to compare placement variants, spacer lengths, or ligand architectures before committing to a preferred build
- Integration with bioorthogonal click chemistry approaches when orthogonal ligation is the best route for selectivity
Focus areas:
Activity retention, manageable purification, reproducible conversion, and better translation from feasibility builds to repeatable conjugate preparation
 Purification & QC Package
Purification & QC Package
Capabilities include:
- Purification planning for oligo-small molecule conjugates using methods appropriate for charge state, hydrophobicity, strand length, and ligand class
- Removal of free ligand, incompletely reacted oligo, truncated sequences, and formulation-related contaminants that can distort downstream studies
- Analytical characterization using orthogonal methods such as LC-MS, RP or ion-pair HPLC, anion-exchange methods, PAGE or CE, and UV/Vis-based label confirmation where relevant
- Optional checks on duplex integrity, buffer exchange, storage handling, and conjugate comparability across candidate builds
- Structured reporting to support internal evaluation, repeat ordering, and follow-on optimization
Deliverables:
Purified conjugates, chromatographic and analytical summaries, identity and purity confirmation, and recommended handling conditions matched to the specific construct
Key Design Parameters for Oligonucleotides Conjugated with Small Molecules
Oligo-small molecule conjugation works best when the scaffold, attachment position, linker, and ligand are designed as a single system. The table below highlights the variables that most often determine whether a construct remains practical for synthesis, purification, and downstream use.
| Design Parameter | Common Options | Development Considerations | Impact on Conjugate Performance | Why It Matters to Customers |
| Oligonucleotide Scaffold | ASO, siRNA, DNA, RNA, aptamer, single-stranded or duplex format | Strand architecture determines tolerated attachment sites and purification behavior | Influences activity retention, hybridization, and feasibility of post-synthetic modification | Helps determine whether the build should be optimized as a probe, delivery construct, or targeting reagent |
| Attachment Position | 5′, 3′, internal, sense-strand, antisense-strand, post-annealing attachment | Placement must avoid blocking functional regions or destabilizing the intended oligo format | Controls steric exposure, duplex behavior, and accessibility of the attached small molecule | Reduces the risk that conjugation improves one property while damaging another |
| Small Molecule Class | Delivery modifier, targeting ligand, affinity label, reporter tag, custom research ligand | Molecule size, polarity, and function affect chemistry selection and cleanup difficulty | Drives uptake behavior, receptor interaction, capture utility, or signal generation | Clarifies whether the project is solving a delivery problem, a targeting problem, or an assay-readout problem |
| Linker and Spacer | PEG-like, alkyl, TEG, short rigid linker, cleavable or non-cleavable connector | Spacer length and polarity must balance presentation, solubility, and synthetic practicality | Affects aggregation risk, target accessibility, and chromatographic separability | Often determines whether a promising design remains workable during purification and storage |
| Reactive Handle | Amino, thiol, azide, alkyne, carboxyl, phosphate-derived coupling handle | Must align with both oligo compatibility and small-molecule functional-group availability | Influences selectivity, conversion efficiency, and side-product profile | Prevents expensive redesign after discovering that the original chemistry route is poorly matched |
| Purification and QC Plan | RP-HPLC, ion-pair HPLC, anion-exchange, desalting, PAGE, CE, LC-MS, UV/Vis | Method choice depends on oligo length, hydrophobicity shift, charge distribution, and label signature | Determines how effectively free ligand and close impurities can be removed and verified | Strong QC planning makes the final conjugate easier to interpret in downstream studies |
Oligo–Small Molecule Conjugation Strategies & Process Development Considerations
There is no single chemistry route that fits every oligonucleotide-small molecule project. Method selection should be driven by the available functional groups, whether the modification is introduced during oligo synthesis or post-synthetically, the sensitivity of the oligo, and how cleanly the final conjugate can be purified and verified.
| Conjugation Strategy | Technical Approach | Common Applications | Development Advantages |
| Pre-installed Modifier Synthesis | Small-molecule-bearing modifiers or phosphoramidite-compatible units are introduced during oligo synthesis | Defined terminal or internal modifications where the ligand chemistry is synthesis-compatible | Strong positional control and streamlined downstream purification for suitable modifier types |
| NHS–Amine Coupling | NHS ester-activated ligands are reacted with amino-modified oligonucleotides to form stable amide bonds | Fluorophores, biotin-type reagents, and small molecules that are conveniently supplied as NHS esters | Straightforward entry route for amino-labeled oligos with broad ligand availability |
| Maleimide–Thiol Coupling | Thiol-modified oligos are coupled to maleimide-bearing ligands or linkers under thiol-selective conditions | Defined terminal attachment, modular linker installation, and custom ligand builds | Useful for chemoselective coupling when controlled sulfur-based handles are available |
| CuAAC or SPAAC Click Chemistry | Azide- and alkyne-bearing partners are joined through copper-catalyzed or copper-free click ligation | Orthogonal ligand attachment, modular screening builds, and sensitive constructs needing selective ligation | High selectivity and flexible handle placement for complex oligo-conjugate design |
| EDC/NHS-Enabled Coupling | Carboxyl-containing ligands are activated to react with primary amines, or related carbodiimide-enabled routes are used where compatible | Small molecules supplied as acids, custom linker installation, and practical amide-bond formation workflows | Expands options when the small molecule is more accessible as a carboxyl-containing intermediate |
| Stepwise Modular Assembly | Linker installation, oligo coupling, duplex formation, and final cleanup are staged rather than done in a single reaction | Duplex oligos, screening sets, and projects with sensitive ligands or multiple build variables | Improves troubleshooting and makes design comparisons easier across several candidate constructs |
Analytical Characterization & Quality Control Framework for Oligo–Small Molecule Conjugates
For oligonucleotide-small molecule conjugates, analytical quality means more than confirming the expected mass shift. It should also show whether the free ligand has been removed, whether the oligo remains intact, and whether the conjugate is practically suitable for the intended delivery, targeting, capture, or assay workflow.
| Analytical Category | Methodology | Purpose in Development | Data Delivered |
| Identity Confirmation | LC-MS or other mass-based confirmation methods where compatible | Verifying successful conjugation and expected molecular composition | Molecular weight confirmation and conjugate identity summary |
| Purity Assessment | RP-HPLC, ion-pair HPLC, anion-exchange HPLC, or related chromatographic methods | Separating product from free ligand, unconjugated oligo, and synthesis-related impurities | Chromatograms, peak interpretation, and purity summary |
| Label-Specific Verification | UV/Vis profiling, fluorescence readout, or ligand-signature checks as appropriate | Confirming the presence of spectroscopic or affinity-related small-molecule features | Absorbance or signal confirmation aligned with the attached label type |
| Oligo Integrity Review | PAGE, CE, duplex assessment, or sequence-format-appropriate integrity checks | Ensuring conjugation and cleanup did not damage the oligonucleotide scaffold | Size or mobility comparison data and integrity observations |
| Handling and Stability Checks | Buffer exchange review, storage observation, or comparative re-analysis where required | Evaluating whether the conjugate remains manageable under intended handling conditions | Recommended storage and handling notes with stability observations |
| Function-Relevant Comparison | Build-to-build comparison of placement, spacer, or ligand variants | Helping select the most practical construct for uptake, targeting, capture, or assay use | Comparative summary supporting design down-selection |
| Documentation Package | Structured reporting of chemistry route, purification, analytics, and recommended conditions | Supporting internal review, repeat builds, and downstream project transfer | Project summary, analytical data package, and handling guidance |
Workflow for Custom Oligonucleotide–Small Molecule Conjugation

We start by reviewing the oligonucleotide format, the small-molecule structure, the intended application, and any existing modifications or analytical constraints. This step helps identify realistic chemistry routes before unnecessary synthesis work begins.
We define the preferred attachment position, linker type, and reactive handle based on the balance required between oligo function, small-molecule presentation, solubility, and purification feasibility.
The conjugation is performed under conditions matched to the selected chemistry. When projects carry more design uncertainty, we can compare a small set of build variants to generate a practical decision point early.
Free ligand, unconjugated oligo, and closely related byproducts are removed using purification methods suited to the construct's charge and hydrophobic profile. This step is critical for meaningful downstream evaluation.
Identity, purity, and construct integrity are assessed using orthogonal analytical methods. Where multiple candidates are prepared, comparative data are organized to support rational selection rather than guesswork.
Final output may include purified conjugates, analytical summaries, and recommended handling conditions, with optional follow-up support for repeat builds, linker refinement, or related conjugation expansion.
Why Choose Our Oligo–Small Molecule Conjugation Platform
We select chemistry routes based on the actual oligo format, strand arrangement, and functional objective, helping reduce the risk of choosing a coupling strategy that is chemically feasible but biologically unhelpful.

Spacer selection is handled as a design variable with practical consequences for uptake, sterics, solubility, purification, and assay behavior, not as a generic add-on made after the chemistry has already been fixed.
Hydrophobic ligands, close-running impurities, and residual free small molecules are common failure points in these projects. We plan purification and QC around those realities so the delivered conjugate is easier to interpret and use.
We support exploratory screening builds, targeted optimization, and repeatable preparation of selected constructs, making it easier to move from concept evaluation to a more stable internal research workflow.
Common Research Applications of Oligonucleotides Conjugated with Small Molecules
Receptor-Targeted Delivery Research
- Ligand-conjugated oligos used to study receptor-mediated uptake and tissue-selective delivery concepts.
- Suitable for evaluating GalNAc-like, folate-like, and other targeting-ligand architectures.
- Useful for comparing site placement and linker effects on uptake-oriented constructs.
Cell Uptake and Trafficking Studies
- Lipophilic or reporter-bearing oligos prepared for internalization, localization, and mechanism-focused studies.
- Often used to compare cholesterol, tocopherol, or related modifiers under matched assay conditions.
- Supports early-stage evaluation of delivery-relevant construct behavior.
Affinity Capture and Pull-Down Reagents
- Biotinylated and other affinity-tagged oligonucleotides used for capture, enrichment, and interaction studies.
- Supports workflows where free-label removal and clean analytical confirmation are especially important.
- Applicable to probe preparation for biochemical and molecular biology research.
Imaging and Assay Probe Development
- Small-molecule-labeled oligos built for fluorescence, signal transduction, and method-development studies.
- Useful for assay optimization, capture-detection workflows, and structure-function comparisons.
- Supports custom probe preparation where labeling site, spacer, and signal behavior must be considered together.
Discuss Your Oligonucleotide–Small Molecule Conjugation Project
Whether you are building a targeted oligo construct, a delivery-modified sequence, or a labeled probe for capture or analytical workflows, we provide technically focused support across strategy design, conjugation, purification, and characterization.
Share your oligonucleotide modality, small-molecule structure, preferred attachment site, and intended application, and we can evaluate a practical chemistry and purification route for your project. If your work spans multiple modification types, we can also coordinate it with related oligonucleotide bioconjugation and specialized conjugation programs.
Frequently Asked Questions (FAQ)
Small molecules can modify the solubility, stability, and hybridization efficiency of oligonucleotides. These modifications are useful for improving binding affinity and enhancing experimental outcomes.
Common methods include chemical reactions such as amide bond formation, click chemistry, and disulfide exchange, which allow precise control over the conjugation process.
Small molecules can increase or decrease hybridization efficiency based on their size, charge, and structure. This can optimize oligonucleotide performance in detection or amplification assays.
Linkers ensure that small molecules are properly positioned to interact with targets while preventing interference with the oligonucleotide's function. The type of linker can impact solubility and stability.

