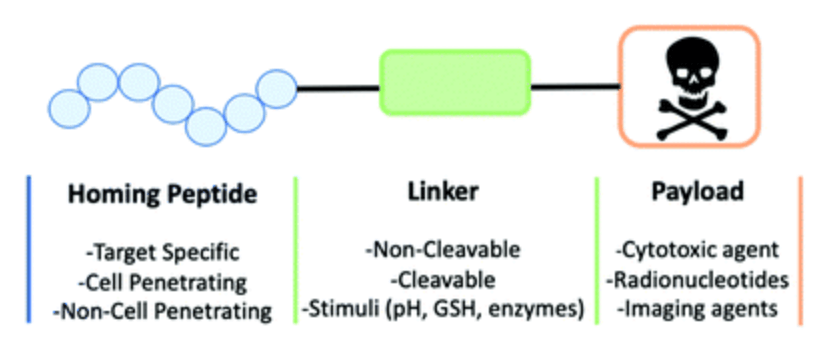
Peptide-Drug Conjugation (PDC)
Site-Selective PDC AssemblyLinker & Payload StrategyDiscovery-to-Preclinical Peptide-Drug Conjugation
Advance targeted payload delivery programs with custom peptide-drug conjugation designed for biotech, pharmaceutical, and CRO teams. Peptide-drug conjugates (PDCs) combine a peptide carrier, a chemical linker, and a bioactive small-molecule payload to create defined constructs for mechanism studies, lead optimization, and preclinical evaluation. As part of our drug conjugation services, we support projects that require careful control of attachment site, linker behavior, payload compatibility, and final conjugate quality.
We integrate peptide design, conjugation route selection, purification, and analytical confirmation into one workflow under our broader peptide conjugation services and custom bioconjugation services. Projects can be tailored for linear, cyclic, PEGylated, or otherwise modified peptides, as well as for payloads whose stability, solubility, or release requirements demand route-specific planning. The goal is not simply to make a bond, but to produce a defined PDC that remains useful for downstream biological interpretation.
What Does Peptide-Drug Conjugation Solve in Real Development Programs?
Peptide-drug conjugation is typically used when a free small molecule does not provide enough selectivity, when intracellular access or tissue localization needs to be improved, or when a project needs a more controllable carrier than a larger biomolecule format. In practice, a PDC must balance four variables at the same time: peptide binding or uptake behavior, linker stability versus release, payload integrity after coupling, and a conjugation site that does not destroy peptide function. A strong PDC strategy therefore starts with the full molecular context rather than a one-size-fits-all coupling method.
Key Challenges in Peptide-Drug Conjugation Projects
A peptide may bind well on its own but lose affinity or internalization efficiency once a bulky or hydrophobic payload is attached. We evaluate terminal versus side-chain attachment, spacer length, and site-selective handle placement to reduce steric interference and preserve peptide function.
One of the most common failure modes in PDC design is a linker that releases the payload too early, or conversely fails to release it under the intended experimental conditions. We help match stable, cleavable, or trigger-responsive linker concepts to the desired exposure and release model.
Many drug payloads reduce aqueous handling, promote aggregation, and broaden chromatographic peaks once coupled to peptides. Our workflows address this through spacer design, sequence-level adjustment where appropriate, reaction-order planning, and purification methods chosen for the actual polarity profile of the final conjugate.
Mixed attachment sites, variable loading, and residual free payload can make biological readouts difficult to interpret. We prioritize defined stoichiometry, site-controlled assembly where feasible, and orthogonal HPLC/UPLC plus LC-MS confirmation so the final material is chemically interpretable before it enters screening.
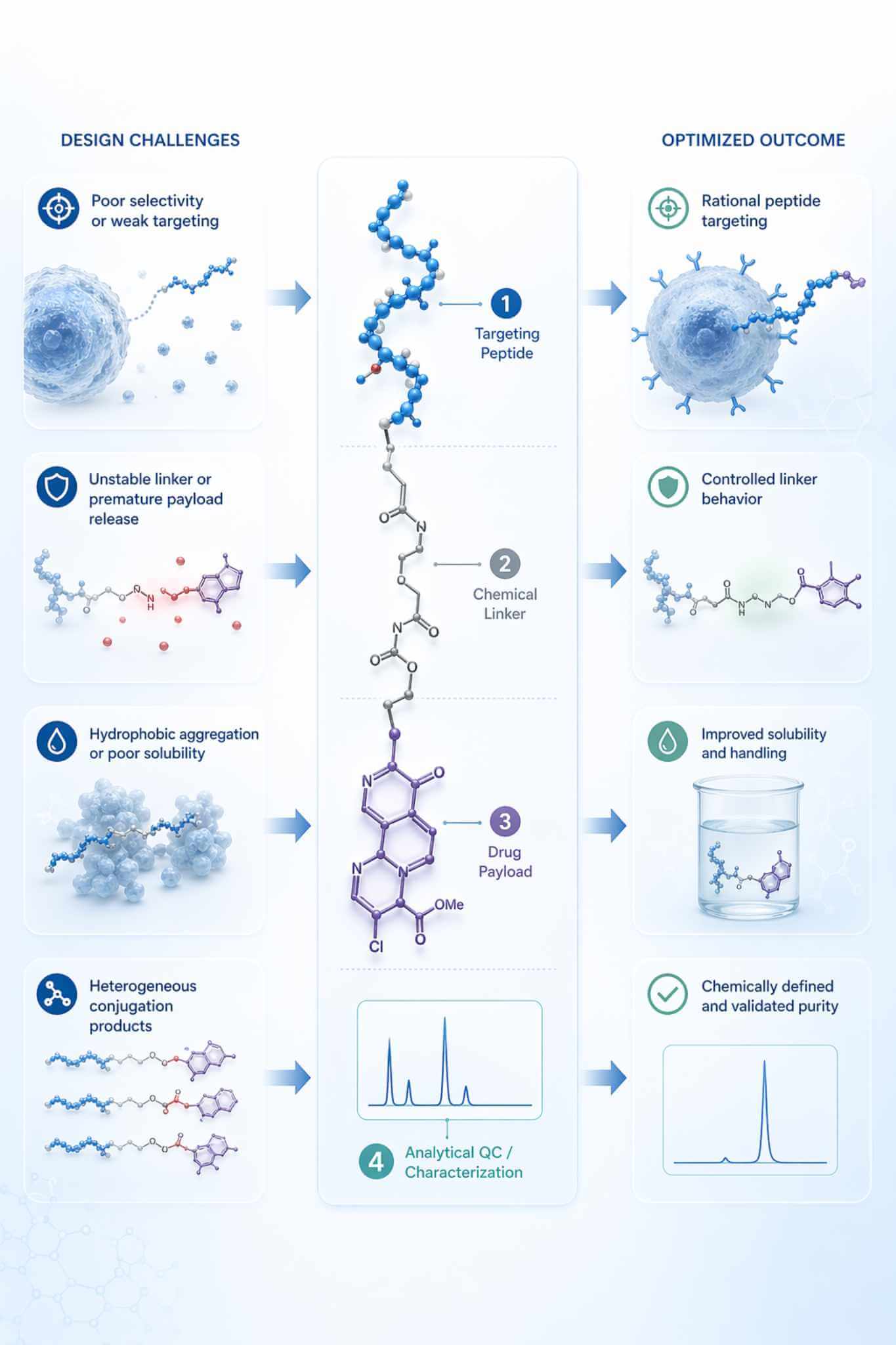 A successful peptide-drug conjugate program depends on balancing peptide function, linker behavior, payload compatibility, and analytical confirmation.
A successful peptide-drug conjugate program depends on balancing peptide function, linker behavior, payload compatibility, and analytical confirmation.
Our Peptide-Drug Conjugation Services
Our PDC support is built around the decision points that matter most in real projects: whether the peptide should target or penetrate, where the payload should be attached, how the linker should behave, how to manage hydrophobicity, and what analytical data are needed to judge success. For programs where a peptide is not the only carrier under consideration, we can also help compare PDC options with related formats such as protein-drug conjugation and small molecule ligand-drug conjugation.
 Peptide Design & Attachment Site Planning
Peptide Design & Attachment Site Planning
Capabilities include:
- Evaluation of targeting peptides, cell-penetrating peptides, shuttle peptides, and modified peptide scaffolds for conjugation feasibility
- Selection of N-terminal, C-terminal, unique Cys, Lys side-chain, or orthogonal handle-based attachment strategies
- Assessment of cyclization, PEG spacing, or sequence-level adjustments to improve stability and handling
- Design review focused on preserving receptor recognition or uptake behavior after conjugation
- Early identification of steric-risk positions and likely sources of product heterogeneity
- Comparison of single-site versus multi-site loading strategies based on project goals
Typical use cases:
Targeted peptide screening, lead format comparison, and early feasibility studies for custom PDC assembly
 Linker & Payload Strategy Development
Linker & Payload Strategy Development
Capabilities include:
- Selection of stable, cleavable, or environment-responsive linker concepts based on the intended release model
- Route planning around payload functional groups, peptide compatibility, and target stoichiometry
- Feasibility assessment for hydrophobic, highly functionalized, or handle-limited payloads
- Spacer and solubilizer recommendations to improve conjugate handling and purification behavior
- Evaluation of maleimide conjugation, click chemistry, and other bioorthogonal reactions when orthogonal assembly is needed
- Payload-first route design to reduce the risk of potency loss during coupling
Typical use cases:
Linker comparison, release tuning, payload replacement, and chemistry de-risking before scale-up
 Custom PDC Synthesis & Purification
Custom PDC Synthesis & Purification
Capabilities include:
- Small-scale route scouting followed by preparative synthesis of selected peptide-drug conjugates
- Integration of peptide synthesis, payload activation or pre-functionalization, conjugation, and cleanup
- Controlled reaction design for site-defined products when the molecule set supports it
- Purification strategies tailored to peptide length, payload hydrophobicity, and linker chemistry
- Isolation of reference materials such as free peptide or free payload when useful for downstream comparison
- Support for quantity progression from feasibility material to larger research batches
Deliverables:
Purified PDC material, yield and purity summary, and route notes aligned with research-stage decision making
 Analytical Characterization & Stability Support
Analytical Characterization & Stability Support
Capabilities include:
- Identity confirmation by LC-MS or HRMS as appropriate for the construct
- Purity profiling by RP-HPLC or UPLC with impurity and residual free component review
- Assessment of product loading, conjugation completeness, and batch-to-batch comparability
- Bench, storage, or fit-for-purpose stress studies to monitor conjugate integrity
- Optional release-focused analytical planning for cleavable linker concepts
- Project-ready reporting to support internal R&D, formulation, and biology teams
Focus areas:
Chemical definition, analytical interpretability, and data packages that help teams move from synthesis to screening with confidence
Key Design Parameters for Peptide-Drug Conjugates
Successful PDC programs depend on aligning peptide biology with chemistry realities. The table below highlights the variables that usually determine whether a conjugate remains selective, synthetically tractable, and analytically well defined.
| Design Parameter | Common Options | What We Evaluate | Impact on the Final Conjugate | Why It Matters to the Project |
| Peptide Role | Targeting peptide, cell-penetrating peptide, shuttle peptide, cyclic scaffold | Binding behavior, internalization profile, and tolerance to chemical modification | Determines where the construct goes and how efficiently it reaches the intended biology | Sets the biological rationale before linker and payload decisions are made |
| Attachment Site | N-terminus, C-terminus, unique Cys, Lys side chain, non-natural amino acid handle | Site selectivity, steric burden, and risk of disrupting peptide activity | Influences product heterogeneity, yield, and retention of peptide function | One of the most important levers for reproducibility and interpretability |
| Linker Behavior | Non-cleavable, enzyme-cleavable, disulfide, acid-labile, self-immolative designs | Stability versus release under the intended experimental conditions | Controls whether the payload stays attached, releases on cue, or degrades too early | Directly affects mechanism readout and structure-activity conclusions |
| Spacer / Solubilizer | PEG spacer, hydrophilic linker, short alkyl spacer, no added spacer | Solubility, purification behavior, and steric separation between peptide and payload | Can improve handling, peak shape, and biological accessibility of the peptide motif | Often decisive for hydrophobic payloads or aggregation-prone constructs |
| Payload Class | Cytotoxic small molecule, enzyme inhibitor, antimicrobial agent, anti-inflammatory scaffold | Functional groups, stability during coupling, and potency retention after modification | Affects route choice, purification difficulty, and final analytical package | Payload chemistry often dictates whether a project is straightforward or high risk |
| Product Definition | Single loading, defined multivalent loading, mixed loading screened in feasibility stage | Stoichiometry control and the expected level of heterogeneity | Influences consistency between lots and quality of downstream biological interpretation | Essential when multiple constructs must be compared side by side |
Conjugation Routes Commonly Used in Peptide-Drug Projects
No single chemistry is universally best for PDC synthesis. The optimal route depends on available functional groups, whether site control is required, how sensitive the payload is, and whether the project prioritizes rapid screening or a highly defined final construct.
| Conjugation Route | Typical Reactive Handles | Strengths | Main Watch-Outs | Best Fit in PDC Work |
| Amide Coupling | Activated carboxyl groups with amines; NHS ester workflows | Robust, broadly accessible, and often easy to implement when handles already exist | Can create heterogeneous products if multiple amines are available on the peptide | Useful for defined terminal attachment or simple feasibility builds |
| Thiol-Maleimide Coupling | Unique cysteine on peptide with maleimide-functionalized payload or linker | Fast and practical route for single-site attachment when a unique thiol is available | Requires attention to thiol availability, exchange side reactions, and hydrolytic stability | Common choice for site-selective PDC assembly and linker screening |
| Cu-Free Click Chemistry | Azide / strained alkyne or tetrazine / TCO pairs | Orthogonal late-stage assembly with good selectivity in multifunctional molecule sets | Requires pre-installed handles and careful control of added linker mass | Well suited to modular peptide, linker, and payload combination studies |
| CuAAC Click Chemistry | Azide / terminal alkyne pairs with copper catalysis | Reliable triazole formation and strong compatibility with modular screening workflows | Copper handling and cleanup may be undesirable for some sensitive payloads | Effective for discovery-stage route development and structure-activity campaigns |
| Oxime or Hydrazone Ligation | Aldehyde or ketone handles with aminooxy or hydrazide partners | Useful when carbonyl chemistry is already built into the molecule design | Reaction rate and stability depend strongly on handle context and pH conditions | Appropriate for specialized payloads or reversible / trigger-linked concepts |
| Disulfide-Based Attachment | Thiol-bearing peptide and reducible sulfur-containing linker systems | Supports redox-sensitive release concepts where reversible attachment is desired | Can be too labile if the project demands high extracellular stability | Useful in release-driven feasibility studies rather than universal platform builds |
Analytical Characterization Framework for Peptide-Drug Conjugates
A PDC is only as useful as the data behind it. Beyond confirming that a conjugation reaction occurred, analytical work should show whether the correct product was made, how much free payload remains, whether the loading is defined, and whether the construct stays intact under the conditions relevant to the project.
| Analytical Attribute | Common Method | What It Confirms | Decision Value |
| Identity Confirmation | LC-MS, HRMS, or deconvoluted mass analysis | Expected molecular weight and presence of the intended conjugated species | Verifies that synthesis produced the right construct before biological testing |
| Purity & Impurity Profile | RP-HPLC or UPLC with UV detection | Relative abundance of the main product, side products, and incomplete reaction species | Helps determine whether a sample is suitable for screening or needs additional purification |
| Free Payload / Free Peptide Review | Orthogonal chromatographic methods and targeted LC-MS checks | Residual unconjugated drug, peptide, or activated intermediate | Prevents misinterpretation of biology caused by carryover of active free components |
| Loading / Stoichiometry | Mass distribution analysis, peak integration, and route-dependent orthogonal calculations | Whether the conjugate is single-loaded, multiply loaded, or mixed | Critical for comparing batches and understanding structure-activity relationships |
| Stability or Release Behavior | Time-course HPLC/LC-MS under storage, buffer, or fit-for-purpose stress conditions | Whether the construct remains intact or releases/degrades under defined conditions | Informs linker choice, storage planning, and downstream assay design |
| Comparability Across Batches | Overlay chromatography, mass comparison, and summary reporting | Consistency in purity, identity, and loading across repeated preparations | Supports reproducible screening campaigns and multi-batch project execution |
Workflow for Custom Peptide-Drug Conjugation

We review peptide sequence or structure, payload identity, available functional handles, target stoichiometry, and intended experimental use. This stage identifies likely route risks early, especially around steric hindrance, hydrophobicity, or unstable handles.
Based on the molecule set, we define attachment site, linker concept, and practical chemistry options. Where appropriate, multiple routes can be compared to balance product definition, speed, and downstream analytical clarity.
Feasibility reactions are used to check coupling efficiency, by-product formation, and handling behavior before committing to larger synthesis. This is especially useful when payload functional groups or peptide sequences create unusual chemistry constraints.
Once the route is selected, the conjugate is synthesized and purified using methods aligned to peptide length, linker composition, and payload polarity. The purification strategy is planned around the actual impurity profile rather than a generic peptide workflow.
HPLC/UPLC and LC-MS are used to confirm identity, purity, and conjugation completeness. When the project requires it, additional stability or release-oriented studies can be added to support linker and payload decisions.
Final deliverables typically include purified material, analytical summaries, and project notes that help biology, formulation, or medicinal chemistry teams continue the program without losing chemical context.
Why Teams Choose Our Peptide-Drug Conjugation Support
We start from the actual handles present on the peptide and payload, then build the route around those constraints. This reduces unnecessary redesign and makes feasibility decisions more practical from the beginning.

Defined attachment sites help reduce heterogeneity, simplify analytics, and improve confidence when comparing conjugates in screening campaigns. We emphasize site-selective approaches whenever the molecule design supports them.
Hydrophobic, unstable, or multifunctional payloads often fail because the route is planned too late. Our service model treats payload compatibility, solubility behavior, and purification burden as core design variables rather than afterthoughts.
We do not stop at synthesis. Our workflows are structured to deliver the identity, purity, loading, and stability information needed to decide whether a PDC is ready for screening, reformulation, or the next round of optimization.
Research Applications of Peptide-Drug Conjugates
Receptor-Directed Payload Delivery
- PDCs can be designed around peptides that recognize overexpressed receptors or cell-surface markers in defined research models.
- This format is useful when a free payload lacks sufficient localization or selectivity on its own.
- Attachment-site planning is particularly important to preserve peptide recognition after coupling.
Cell-Penetrating Peptide Payload Programs
- Cell-penetrating peptides can help move selected small molecules across difficult cellular barriers in mechanistic studies.
- These programs often require careful balance between uptake benefit and unwanted nonspecific interaction.
- Linker and stoichiometry choices strongly influence interpretability in intracellular delivery work.
Tissue-Homing or Shuttle Peptide Studies
- Peptides used as tissue-homing or transport motifs can be paired with payloads when larger carrier systems are not ideal.
- These projects often focus on uptake, localization, and exposure differences between conjugated and unconjugated molecules.
- Spacer choice is frequently important to avoid masking the peptide recognition element.
Anti-Inflammatory and Fibrosis-Related Research
- PDC strategies can be explored for targeted delivery of small molecules used in inflammatory or fibrotic pathway research.
- The emphasis is usually on improving localization or reducing the free-exposure profile of the unconjugated scaffold.
- Stable linker concepts are often favored when intact conjugate exposure is the design objective.
Antimicrobial and Anti-Infective Conjugate Research
- Peptide carriers can also be explored with antimicrobial or anti-infective small molecules when targeted delivery or uptake is under investigation.
- These programs often require early review of payload stability and analytical detectability.
- Defined stoichiometry is especially useful when comparing multiple constructs in screening.
PDC Design-Space and SAR Optimization
- Many teams use PDC synthesis to compare peptide variants, attachment sites, linker types, or payload classes in parallel.
- This application benefits from consistent purification and a comparable analytical framework across all constructs.
- For background reading, teams often start with current overviews such as advances in peptide-drug conjugates.
| Name | TTP | Payload | Linker | Indication | Development phase |
| ANG1005 | Angiopep-2 | Paclitaxel | Succinic acid | Leptomeningeal metastases Glioma Glioblastoma brain tumor, recurrent Breast cancer brain metastases Advanced solid tumors with and without bain metastases | Phase III Phase II Phase II Phase I |
| GRN1005 | Angiopep-2 | Paclitaxel | Succinic acid | Breast cancer brain metastases; non-small cell lung cancer (nsclc) with brain metastases | Phase II |
| BT1718 | MT1-MMP binder | DM1 | Disulfide | Advanced solid tumours non-small cell lung cancer non-small cell lung sarcoma oesophageal cancer | Phase I/II |
| BT5528 | EphA2 binder | MMAE | Amide | Solid tumours EphA2-positive NSCLC | Phase I |
| BT8009 | Nectin-4 binder | MMAE | Amide | Solid tumors | Phase I |
| TH1902 | TH19P01 | Docetaxel | Succinic acid | Solid tumors | Phase I |
| TH1904 | TH19P01 | Doxorubicin | Succinic acid | Solid tumors | |
| G-202 (mipsagargin) | DgEgEgEgE | Thapsigargin | Amide | Solid tumors | Phase II |
| NGR015 (NGR-hTNF) | CNGRCG(1,5 SS) | hTNF | Amide | Malignant pleural mesothelioma | Phase III |
| tTF-NGR | GNGRAHA | tTF | Amide | Malignant solid tumors lymphomas | Phase I |
| PEN-221 | fCYwKTCC (2,7 SS) | DM-1 | Disulfide | Neuroendocrine tumors arcinoma, small cell lung | Phase I/II |
| CBP-1008 | CB-20BK | MMAE | Amide | Advanced solid tumor | Phase I |
| CBP-1018 | LDC10B | MMAE | Amide | Lung tumor | Phase I |
| SOR-C13 | folate | MMAE | Amide | Advanced malignant solid neoplasm | Phase I |
| Melflufen (delisted) | Flufenamide | Melphalan | AcOH | Multiple myeloma | Approved for marketing |
| 177Lu-dotatate (Lutathera) | Tyr-3-octreotate | 177Lu | DOTA | Neuroendocrine tumors | Approved for marketing |
| 177Lu-PSMA-617 | Glu-urea-R | 177Lu | DOTA | Prostate cancer | Phase I |
| [18F]AlF-NOTA- octreotide | octreotide | 18F | NOTA | PET or GEP-NETs; Neuroendocrine tumors | Phase I/II/III |
| [18F]Fluciclatide | RGD | 18F | PEG | PET imaging | Phase II |
| [18F]RGD-K5 | cyclo(RGDfK) | 18F | NOTA | PET imaging | Phase II |
| 68Ga-NODAGA-E[cyclo(RGDyK)]2 | E[cyclo (RGDyK)]2 | 68Ga | NODAGA | PET imaging | Phase II |
| 68Ga-NOTA-BBN-RGD | cyclo(RGDyK) and BBN | 68Ga | NOTA | PET/CT and PET imaging | Phase I |
| 90Y-DOTATOC | 3Tyr-octreotate | 90Y | DOTA | PRRT | Phase II |
| 99mTc-3PRGD2 | 3Tyr-octreotate | 99mTc | 3PRGD2 | Breast cancer SPECT/CT scan | Phase I |
| 111In-DTPA-octreotide | 3Tyr-octreotate | 111In | DTPA | Brain and central nervous system Tumors PET imaging | Phase I |
Peptide-drug conjugates in clinical trials and approved for marketing. (Fu, Chen; et al.2022)
Build a Peptide-Drug Conjugation Strategy Around Your Actual Molecule Set
Whether you are comparing peptide carriers, screening linker options, or troubleshooting a difficult payload, our team can help translate your peptide and small-molecule inputs into a defined conjugation plan with practical chemistry and fit-for-purpose analytics.
We support discovery and preclinical PDC programs that need more than a generic coupling reaction — including route selection, attachment-site control, purification strategy, and analytical confirmation aligned with downstream research decisions. Contact our scientific team to discuss your peptide-drug conjugation project and receive a tailored plan for peptide, linker, payload, and analytical workflow design.
Frequently Asked Questions (FAQ)
Peptide-drug conjugation involves attaching a peptide to a small molecule, typically to enhance the stability, solubility, or specificity of the drug. This technology is widely used in research to create targeted delivery systems, improve molecular interactions, and explore protein-ligand relationships in various biological studies.
Common conjugation methods include covalent binding through click chemistry, thiol-maleimide reactions, and amide bond formation. These methods are selected based on the desired stability, solubility, and efficiency of the conjugate, and are tailored to suit specific research needs, from protein targeting to drug solubility improvement.
By attaching drugs to peptides, the resulting conjugates often exhibit improved solubility, stability, and longer shelf life. The peptide acts as a stabilizer, preventing degradation and enhancing the pharmacokinetic profile, which is essential for studying compound efficacy and optimizing drug delivery.
The main challenges include ensuring efficient conjugation without compromising peptide activity, optimizing the drug-to-peptide ratio (DAR) for stability, and ensuring that the conjugates perform consistently under different experimental conditions.

