Aptamer Liposome Conjugation
Aptamer liposome conjugates can greatly improve the targeting efficiency of drug-loaded liposomes to tumor cells, achieve precise drug delivery and reduce toxicity. As a leading CRO, BOC Sciences is committed to providing custom liposome conjugation services. We have the ability to conjugate aptamers with liposomes to provide innovative solutions for drug delivery.
Aptamer Conjugated Nanoparticles
Aptamers are short, single-stranded DNA or RNA oligonucleotide molecules with a distinct 3D structure, which can be created by systematic evolution of ligands by exponential enrichment (SELEX). The precision of aptamers in recognizing and binding to molecular targets has positioned them as ideal candidates for guiding drug-loaded nanoparticles to their intended destinations within the body.
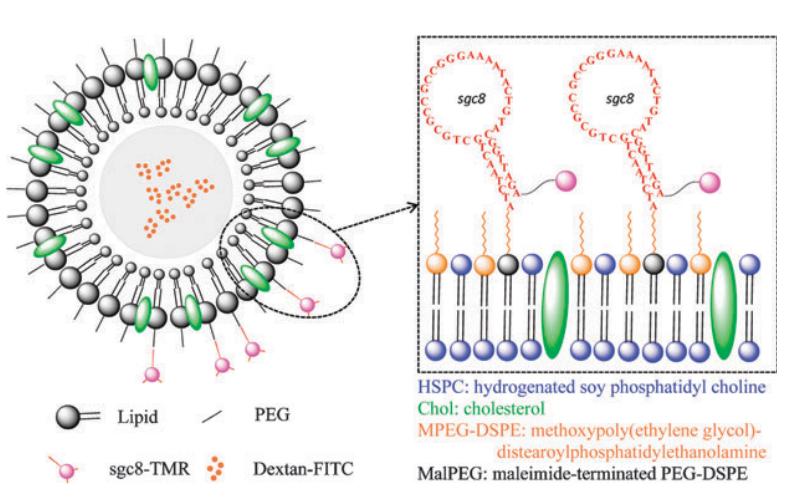 Fig. 1 Aptamer liposome conjugation for targeted delivery (Kang, 2010)
Fig. 1 Aptamer liposome conjugation for targeted delivery (Kang, 2010)
Lipid bilayer vesicles made of cholesterol and phospholipids dissolved in water are called liposomes, and they are one of the most widely used formulations for lowering drug toxicity and enhancing targeting. The non-polar groups are joined to each other to form a bilayer, so that the inside of the bilayer and the vesicles can encapsulate fat-soluble drugs and water-soluble drugs, respectively, thus playing the role of drug carriers. Surface modification of liposome nanoparticles using aptamers is a common approach to identify cancer cell surface targets. Enables the targeted delivery of therapeutic agents such as doxorubicin and docetaxel to cancer cells to treat tumors without affecting normal cells.
Our Services for Aptamer-based Liposome
BOC Sciences uses state-of-the-art conjugation technology to ensure successful integration of aptamers onto the liposome surface. Furthermore, we are capable of encapsulating a wide range of active substances into liposomes, such as small compounds, peptides, proteins, nucleic acid aptamers, etc.
1. Aptamer Synthesis and Modification
Design aptamers that specifically recognize the desired cells or tissues. Aptamers are often modified to enhance their stability and binding affinity. Common modifications include the addition of thiol groups and biotin.
2. Liposome Preparation and Activation
Common lipids include phospholipids such as phosphatidylcholine and cholesterol. Use established methods like thin-film hydration, sonication, or extrusion to form liposomes with the desired size and characteristics. Add functional groups like maleimide, amino, or carboxyl groups to the liposomal surface so they can interact with the modified aptamers.
3. Aptamer Liposome Conjugation
Conjugating the functional groups on the liposome surface with the modified groups on the aptamer. If necessary, add a crosslinker to facilitate the reaction, such as succinimidyl-4-(N-maleimidomethyl) cyclohexane-1-carboxylate (SMCC) for maleimide chemistry.
4. Aptamer-based Liposome Characterization
- Confirmation of Conjugation: Use techniques like gel electrophoresis or mass spectrometry to confirm the successful conjugation of aptamers to liposomes.
- Size and Zeta Potential Analysis: Characterize the size and surface charge of the aptamer-liposome conjugates using dynamic light scattering (DLS) and zeta potential measurements.
- Binding Affinity: Assess the binding affinity of the aptamer-liposome conjugates to the target biomarkers through in vitro assays.
- Cellular Uptake: Evaluate the cellular uptake of the conjugates using cell culture models to ensure effective targeting.
Advantages of Aptamers
Aptamers can form a specific three-dimensional spatial configuration by folding secondary and tertiary structures, and then bind to different target molecules with high specificity and affinity by van der Waals forces, hydrogen bonding, electrostatic interactions, base stacking forces, etc. Aptamers are characterized by wide range of target molecules, high stability, non-immunogenicity, easy penetration into tissues, low price, easy synthesis and modification, etc. They have broad application prospects in biomarker discovery, cancer diagnosis and targeted drug delivery system development.
Applications of Aptamer-Functionalized Liposomes
Aptamer-functionalized liposomes harness these advantages, combining the high specificity of aptamers with the drug encapsulation capabilities of liposomes. Recent years, aptamers screened with some clinically important tumor markers or biomarkers as targets have gained rapid development in tumor targeting research. For example, nucleolin ( NCL, C23 ) is highly expressed in breast cancer cell membranes and its specific aptamer AS1411 is often used for breast cancer targeting liposome studies. The aptamer AS1411 modified liposome conjugates can enhance the killing effect on breast cancer cells and improve the antitumor effect on breast cancer xenografts when compared with normal liposomes.
Our Advantages
- Synthesis and screening of specific aptamers
- Extensive expertise in liposomes
- Various conjugation technologies combine aptamers and liposomes
- Liposome encapsulation based on cargo or release conditions
Frequently Asked Questions (FAQ)
Where the aptamer is positioned on the liposome surface can change how well it binds to targets. Proper spacing improves the chances that multiple aptamers can engage simultaneously.
Yes. Liposome membrane properties, such as flexibility or rigidity, can affect how aptamers fold and how accessible they are for binding.
Changes in temperature, pH, or ionic strength can alter aptamer shape and accessibility, impacting binding performance.
Yes. Controlling how many aptamers are on each liposome allows fine-tuning of binding strength and efficiency without changing the aptamer sequence.
Using strategic placement and spacing, researchers can enhance multivalent interactions, ensuring effective molecular recognition in experiments.


Reference
- Kang, H., et al., A liposome-based nanostructure for aptamer directed delivery, Chem. Commun., 2010, 46, 249-251.