Amino Acid Conjugation
Site-Aware Amino Acid Coupling StrategiesPEG, Probe, Lipid, and Small-Molecule Conjugation Support
Amino acid conjugation is a practical route for turning a simple amino acid or amino acid derivative into a more useful research intermediate, labeled building block, or functional conjugate. For project teams working in chemical biology, peptide science, assay development, and bioconjugation, the value is not just forming a new bond—it is choosing a coupling strategy that preserves the reactive handle you still need, controls where the modification happens, and generates a product that can be purified and confirmed without ambiguity.
Our amino acid conjugation services support custom attachment of PEG chains, fluorophores, biotin, lipids, drugs, haptens, spacers, and other functional moieties to natural, protected, or non-natural amino acids. We design routes around the actual chemistry of your substrate—such as the α-amino group, α-carboxyl group, or side chains including lysine, cysteine, tyrosine, aspartic acid, and glutamic acid—and align the work with downstream use cases such as peptide synthesis, probe generation, biotinylation, fluorescence labeling, or broader protein/peptide crosslinking.
What Is Amino Acid Conjugation?
Amino acid conjugation is the covalent attachment of an amino acid, amino acid derivative, or amino-acid-based building block to another functional entity in order to introduce a specific property or synthetic handle. Depending on project goals, the conjugated partner may be a polymer, fluorophore, affinity tag, lipid tail, drug-like fragment, linker, solid-support handle, or another biomolecule. In practice, successful amino acid conjugation depends on matching the right reactive group pair, controlling chemoselectivity, and using a linker architecture that improves the final construct without compromising later synthesis, purification, or assay performance. This is why amino acid conjugation is often used to improve solubility, install analytical labels, create orthogonal intermediates, or build customized starting materials for peptide and bioconjugation workflows.
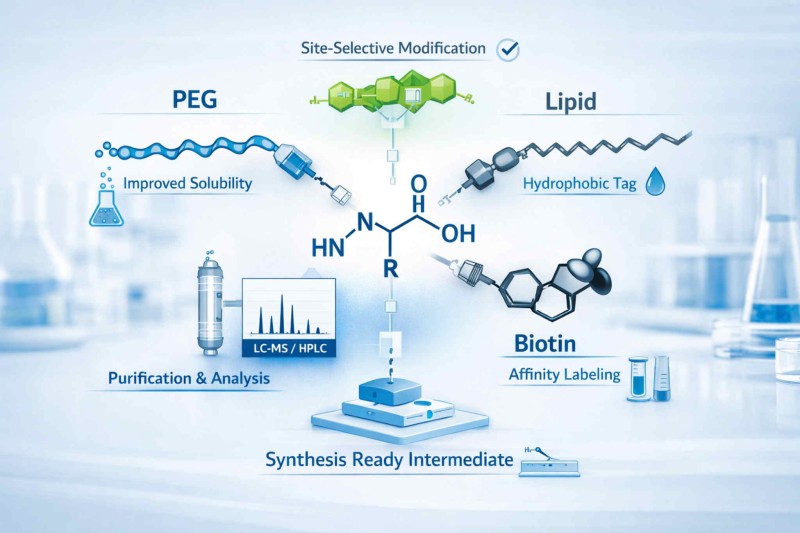 Amino acid conjugation can be tailored to install solubilizing, labeling, affinity, or lipid handles while preserving the chemistry needed for downstream synthesis and analysis.
Amino acid conjugation can be tailored to install solubilizing, labeling, affinity, or lipid handles while preserving the chemistry needed for downstream synthesis and analysis.
Real Development Problems Amino Acid Conjugation Can Help Solve
Hydrophobic fragments, dyes, lipids, and certain drug-like moieties can be hard to dissolve, transfer, or integrate into aqueous workflows. Amino acid conjugation can introduce polar functionality, a defined spacer, or a more synthesis-friendly intermediate that makes downstream reactions and analytical work more manageable.
When multiple reactive groups are present, uncontrolled modification can generate mixtures or block the handle you intended to keep free. We help define whether the optimal entry point is the N-terminus, C-terminus, lysine side chain, cysteine thiol, phenolic handle, or a preinstalled orthogonal group so the final conjugate remains fit for its intended use.
Many clients do not need "a conjugate" in the abstract—they need one that still works in SPPS, fragment coupling, click-functionalization, resin loading, or later deprotection steps. Our route design considers orthogonality, linker stability, and the order of operations so the conjugate can move cleanly into the next stage of the project.
Amino acid conjugation projects often produce partially reacted materials, regioisomeric species, residual activated reagents, or hydrolyzed byproducts. We build purification and analytical strategy into the plan early so the final material is not only synthesized, but also distinguishable from look-alike impurities by suitable chromatographic and spectrometric methods.
Discovery programs frequently stall when a custom amino acid derivative exists only as a small crude sample with incomplete characterization. We focus on deliverables that are usable in real workflows, with identity confirmation, purity assessment, and documentation matched to the stage and purpose of the material.
A conjugate that works once at exploratory scale may fail when reproduced by another team or carried forward to a larger batch. We emphasize route clarity, controlled reaction conditions, and fit-for-purpose analytical comparison so follow-on synthesis has a cleaner starting point.
Our Amino Acid Conjugation Services
We support amino acid conjugation as a standalone chemistry service or as part of a broader peptide, labeling, or bioconjugation program. Service scope is defined around the actual objective of the project—building a functional intermediate, improving molecular handling, introducing a tracking tag, preparing a lipidated derivative, or generating analytically verified material for downstream studies.
 Amino Acid–PEG Conjugation
Amino Acid–PEG Conjugation
Capabilities include:
- PEG attachment to amino, carboxyl, thiol, or preinstalled orthogonal handles on amino acid substrates
- Support for linear or functionally activated PEG reagents, selected according to the target handle and intended downstream chemistry
- Linker and spacer design to balance solubility improvement with synthetic practicality
- Route planning for protected amino acid intermediates used in peptide assembly or follow-on derivatization
- Purification and LC-MS/HPLC confirmation of the final PEGylated amino acid
Typical use cases:
Solubility tuning, spacer installation, modified building blocks for peptide chemistry, and intermediate preparation for more complex conjugation workflows
 Probe, Dye, and Affinity-Tag Conjugation
Probe, Dye, and Affinity-Tag Conjugation
Capabilities include:
- Conjugation of amino acids with fluorophores, chromophores, biotin, quencher groups, and related affinity or tracking handles
- Selection of coupling chemistry based on handle availability, desired stability, and analytical readout
- Preparation of intermediates that can support downstream biotinylated amino acid workflows or broader fluorescence labeling projects
- UV/Vis or fluorescence confirmation when applicable, alongside LC-MS and chromatographic purity assessment
Typical use cases:
Analytical probes, pull-down reagents, labeled peptide precursors, and assay-oriented custom building blocks
 Lipidated and Hydrophobic-Tail Amino Acid Conjugates
Lipidated and Hydrophobic-Tail Amino Acid Conjugates
Capabilities include:
- Attachment of fatty acids, hydrophobic tails, or membrane-interacting groups to selected amino acid substrates
- Consideration of chain length, linker type, and attachment site to preserve synthetic accessibility
- Support for N-acylation, side-chain modification, or linker-first strategies where appropriate
- Purification planning for highly hydrophobic products and mixed polarity systems
Typical use cases:
Lipoamino acids, membrane-active intermediates, self-assembly studies, and customized building blocks for peptide or delivery-oriented constructs
 Small-Molecule and Linker Installation
Small-Molecule and Linker Installation
Capabilities include:
- Conjugation of amino acids with drug-like fragments, haptens, cleavable or non-cleavable linkers, and custom spacers
- Feasibility review of functional group compatibility before route selection
- Design of protected intermediates that preserve a downstream coupling position
- Support for building blocks used in broader drug conjugation services or custom peptide modification projects
Typical use cases:
Custom linker-bearing amino acids, hapten intermediates, synthetic probes, and molecular redesign projects that require a defined amino-acid-based entry point
 Protected, Non-Natural, and Orthogonal Amino Acid Derivatives
Protected, Non-Natural, and Orthogonal Amino Acid Derivatives
Capabilities include:
- Work with protected amino acids and derivatives where orthogonality is critical to the synthesis plan
- Installation of azide, alkyne, maleimide-compatible, thiol-reactive, or other custom handles according to project needs
- Preparation of specialty intermediates relevant to click-ready and modular conjugation workflows
- Optional alignment with stable isotope labeling of amino acids & derivatives when tracer or analytical applications are part of the design
Typical use cases:
Orthogonal synthesis, modular building block development, structure–activity studies, and bespoke research reagents
 Purification, Characterization, and Batch Support
Purification, Characterization, and Batch Support
Capabilities include:
- Preparative purification strategy based on polarity, hydrophobicity, and byproduct profile
- Identity confirmation by LC-MS and other suitable structural methods as applicable
- Purity assessment by HPLC, UPLC, or fit-for-purpose chromatographic methods
- Comparison of repeat batches when projects move beyond one-off synthesis
- Technical reporting that makes the material easier to transfer into downstream research workflows
Typical use cases:
Clients who need characterized custom material rather than exploratory crude products, especially for follow-on peptide synthesis or analytical method development
Common Design Routes for Amino Acid Conjugation
The best route is usually determined by the handle you must preserve, the one you want to react, and the downstream conditions the conjugate must survive. The table below summarizes practical design patterns often considered in amino acid conjugation projects.
| Project Goal | Typical Amino Acid Handle | Common Partner Handle | Representative Chemistry | Key Development Watchpoint |
| Build a Stable Labeled Intermediate | α-Amino group or lysine side chain | Activated ester, isothiocyanate, or preactivated dye/tag | Amide-forming or amine-reactive coupling | Avoid over-reaction when multiple amines are present and confirm labeling position where it matters |
| Prepare a Thiol-Directed Conjugate | Cysteine thiol or a thiol-bearing derivative | Maleimide or other thiol-reactive linker | Thioether-forming conjugation | Control oxidation state and evaluate whether the linkage stability matches the intended workflow |
| Introduce a Solubilizing Spacer | Amino, carboxyl, or orthogonal handle on a protected amino acid | Activated PEG or spacer reagent | Solution-phase coupling or staged linker installation | Choose chain length and protecting strategy that improve handling without blocking later synthesis |
| Create a Click-Ready Building Block | Preinstalled azide or alkyne functionality | Complementary azide/alkyne partner | Copper-catalyzed or copper-free click-compatible route | Ensure orthogonality with other functional groups and assess whether metal-free conditions are needed |
| Generate a Lipidated Derivative | N-terminus, lysine side chain, or custom spacer position | Activated fatty acid or hydrophobic tail | N-acylation or linker-mediated coupling | Balance hydrophobicity with purification feasibility and verify the product remains usable in downstream synthesis |
| Preserve One Handle for Later Assembly | Selectively protected amino or carboxyl group | Linker, tag, or small-molecule fragment | Orthogonal protection/deprotection sequence | The order of operations is often more important than the final coupling step itself |
Conjugation Strategy and Process Development Considerations
Amino acid conjugation is rarely a one-reagent problem. In practice, route success depends on how the substrate is protected, how many competing nucleophiles or electrophiles are present, whether the product must enter peptide synthesis or labeling workflows later, and how easily the desired product can be separated from closely related species.
| Development Variable | Common Options | Why It Matters | Project Impact |
| Starting Material State | Free amino acid, protected amino acid, amino acid ester, resin-bound intermediate | Determines which handle is accessible and whether the route should be run in solution or integrated into a stepwise synthesis workflow | Strongly affects selectivity, workup, and later deprotection planning |
| Reactive Group Pairing | Amine-reactive, thiol-reactive, carboxyl-activation, click-compatible, aldehyde/ketone-directed | Different pairings offer different balances of speed, chemoselectivity, and byproduct complexity | Influences conversion, heterogeneity, and compatibility with sensitive substrates |
| Attachment Site | N-terminus, C-terminus, lysine, cysteine, tyrosine, or engineered orthogonal handle | Site selection controls whether the conjugate keeps the functional handle needed for the next step | Affects usability of the final building block and the risk of unintended side products |
| Linker Architecture | Direct attachment, short spacer, PEG spacer, hydrophobic tail, cleavable segment | The linker can change solubility, steric accessibility, and downstream compatibility | Often determines whether the final conjugate behaves well in synthesis and assay settings |
| Purification Route | Recrystallization, precipitation, flash chromatography, prep-HPLC, orthogonal cleanup sequence | Closely related byproducts can make purification the true bottleneck of the project | Directly impacts isolated purity, recovery, and batch reproducibility |
| Scale Strategy | Exploratory milligram batch, confirmed repeat batch, larger custom preparation | A route that works once may require adjustment before repeatable delivery | Impacts reagent choice, handling risk, impurity control, and documentation expectations |
Analytical Characterization for Amino Acid Conjugates
Analytical confirmation is especially important in amino acid conjugation because many projects involve small structural changes with large functional consequences. The most useful analytical package is typically the one that proves the intended structure, identifies likely side products, and gives the client enough confidence to move the material into the next workflow.
| Analytical Category | Typical Methods | What It Confirms | When It Is Especially Valuable |
| Identity Confirmation | LC-MS or HRMS, and other structural methods as applicable | Expected molecular weight and evidence for successful conjugation | Essential for nearly all custom amino acid conjugation projects |
| Purity Assessment | HPLC, UPLC, or orthogonal chromatographic methods | Relative purity and visibility of partially reacted or hydrolyzed species | Critical when the material will be used as a downstream synthetic intermediate |
| Structural Support | NMR and related spectroscopic methods where suitable | Bond formation pattern, protecting group state, and structural consistency | Helpful for linker-bearing, protected, or more complex small-molecule conjugates |
| Optical or Functional Readout | UV/Vis, fluorescence, or other application-linked measurements | Presence of the installed probe or labeled moiety | Important for dye-tagged or affinity-oriented conjugates |
| Hydrophobicity / Retention Behavior | Comparative chromatographic profiling | Whether linker, PEG, or lipid installation changed retention as expected | Useful for PEGylated, lipidated, or mixed-polarity derivatives |
| Batch Comparison | Overlay of chromatograms and mass data across repeat preparations | Reproducibility of the selected route | Valuable when the project moves beyond an initial feasibility batch |
Our Amino Acid Conjugation Workflow

We review the starting structure, target conjugate, available reactive handles, scale expectations, and downstream use. This step clarifies whether the project is best approached as direct amino acid modification, protected intermediate synthesis, staged linker installation, or a broader custom conjugation program.
We identify the most appropriate conjugation position and reactive group pair based on chemoselectivity, functional group compatibility, and the handle that must remain available after the reaction. At this stage, we also consider linker length, protection strategy, and likely impurity risks.
A fit-for-purpose route is developed and tested at exploratory scale. This allows early assessment of conversion, workup behavior, purification difficulty, and whether the chosen strategy is delivering the intended product rather than a mixture of closely related species.
Purification is selected according to polarity, stability, and impurity profile. Identity and purity are then confirmed using appropriate analytical methods so the material is suitable for downstream synthesis, labeling, screening, or assay use.
If follow-on quantities are required, the route can be refined for better robustness, cleaner impurity control, and more reproducible isolation. This step is particularly useful for clients who need the same amino acid conjugate more than once or want a smoother transfer into a larger program.
Final deliverables are provided with the level of analytical support appropriate to the project scope, helping internal chemistry, peptide, or assay teams use the material more efficiently in subsequent work.
Why Clients Choose Our Amino Acid Conjugation Support
We do not treat amino acid conjugation as a generic labeling exercise. Route design is based on your substrate, your intended attachment site, and the handle that still needs to function after conjugation.

Many projects require more than an isolated product—they require a conjugate that remains compatible with peptide synthesis, deprotection, click assembly, or later derivatization. We account for that during route planning.
Competing reactive groups, partial conversions, and closely related side products are common in amino acid conjugation. We emphasize practical selectivity and analytical discrimination so the product profile is understood, not assumed.
Our focus is on delivering amino acid conjugates that can move into the next step with fewer surprises, supported by appropriate purification, structure confirmation, and transfer-ready project information.
Typical Application Areas for Amino Acid Conjugation
Peptide and Peptidomimetic Building Blocks
- Preparation of modified amino acids for SPPS or fragment-based assembly.
- Installation of spacers, tags, or hydrophobic groups before peptide construction.
- Creation of custom intermediates for structure–activity exploration.
Fluorescent, Biotin, and Assay-Oriented Probes
- Labeled amino acid derivatives for pull-down, tracing, or method development.
- Customized probe precursors for assay construction and validation.
- Affinity-tagged intermediates that connect chemistry and bioassay workflows.
Solubility and Spacer Optimization
- PEGylated or spacer-bearing amino acids that improve material handling.
- Support for molecules that are difficult to dissolve, transfer, or purify in their parent form.
- Conjugation design that makes subsequent synthesis steps more manageable.
Lipidated and Membrane-Interacting Constructs
- Fatty-acid-bearing amino acids and related hydrophobic derivatives.
- Intermediates for self-assembly, membrane interaction, or lipopeptide-oriented studies.
- Route support for products where polarity balance is a central challenge.
Small-Molecule Derivatization and Linker Studies
- Attachment of amino acid units to fragments, haptens, or linker-bearing molecules.
- Custom design of conjugates used to compare spacer length, polarity, or attachment mode.
- Preparation of intermediates that support broader bioconjugation strategy work.
Specialty Analytical and Isotope-Related Workflows
- Amino-acid-based standards or specialty derivatives used in analytical development.
- Support for projects that combine conjugation design with isotope-labeled starting materials.
- Materials intended for structure confirmation, tracking, or comparative method work.
Start Your Amino Acid Conjugation Project with a Route Built for the End Use
Whether you need a PEGylated amino acid, a labeled derivative, a lipidated intermediate, or a custom linker-bearing building block, we can design an amino acid conjugation strategy around the actual chemistry and downstream purpose of the material.
We work with research teams that need more than a nominal synthesis result: they need a conjugate that can be purified, confirmed, and transferred into the next experimental stage with fewer delays.Contact our scientific team to discuss your amino acid conjugation project, target structure, and preferred deliverables.
Frequently Asked Questions (FAQ)
Commonly conjugated amino acids include glycine, glutamine, taurine, ornithine, and serine, which help neutralize and solubilize various metabolites and xenobiotics.
Glycine frequently conjugates with carboxylic acids to form hippuric acid-like derivatives, aiding in the excretion of toxic substances like benzoic acid.
Amino acid conjugation occurs in phase II metabolism, where it facilitates the excretion of metabolites generated during phase I.
By attaching polar amino acids to hydrophobic molecules, conjugation increases their water solubility, enabling efficient elimination from the body.
It detoxifies potentially harmful compounds, protects cellular integrity, and supports the body's ability to metabolize and excrete drugs and endogenous substances.

