Peptide RNA Conjugation
Defined Peptide–RNA ArchitecturesRNA-Compatible Linker & Handle DesignPurified Conjugates for Delivery & Functional Studies
Build research-ready peptide–RNA conjugates with a workflow designed for teams working in RNA delivery, intracellular trafficking, molecular recognition, encoded library technologies, and custom assay development. Peptide RNA conjugation combines the sequence-driven function of RNA with the targeting, uptake-enhancing, membrane-interacting, or assembly roles of peptides, making it a practical format when unconjugated RNA is difficult to deliver, unstable in the working system, or not easy to evaluate in a defined covalent construct.
We support custom development from RNA format review and peptide selection through reactive handle planning, conjugation route design, purification, and analytical characterization. Projects can be aligned with broader oligonucleotide bioconjugation needs and coordinated with related peptide oligonucleotide conjugation or peptide conjugation services when a program includes multiple conjugate formats.
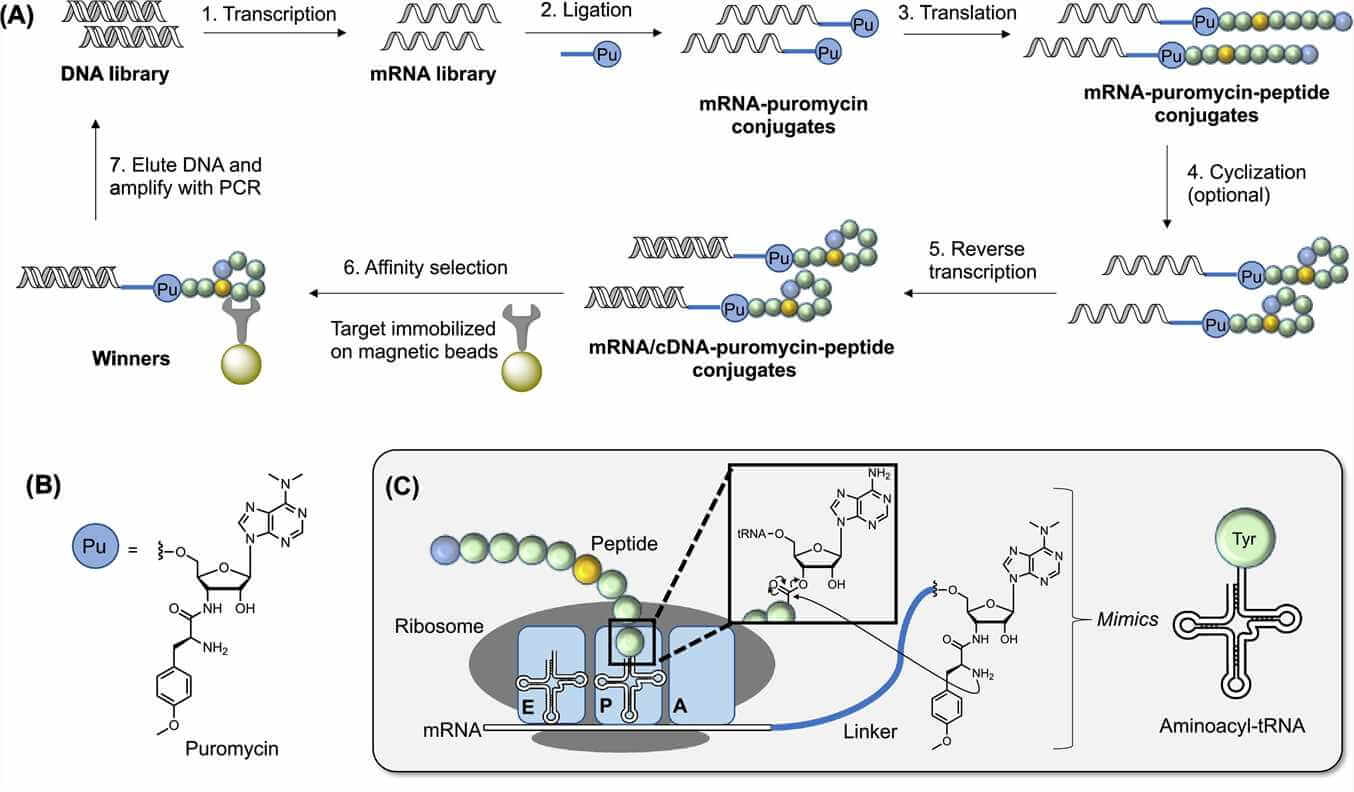 Peptides-RNA conjugates in mRNA display. (Peacock. 2021)
Peptides-RNA conjugates in mRNA display. (Peacock. 2021)
What Problems Can Peptide RNA Conjugation Solve?
Many RNA projects stall not because the sequence concept is weak, but because the unconjugated construct enters cells inefficiently, is cleared or degraded during handling, becomes trapped in endosomal compartments, or cannot be evaluated in a controlled molecular format. Peptide RNA conjugation is used to convert an RNA payload into a defined hybrid construct that can support uptake studies, receptor-focused delivery research, intracellular trafficking analysis, encoded selection workflows, or mechanistic comparison of linker and attachment-site effects. In practice, this helps research teams address common gaps such as poor cellular exposure, unstable noncovalent complex formation, loss of duplex behavior after modification, difficult purification of partially reacted material, and limited analytical confidence when moving from concept screening to reproducible build cycles.
A useful conjugation strategy considers RNA class, peptide role, attachment site, linker stability, charge balance, purification behavior, and downstream assay conditions together rather than as isolated variables. That is especially important when the same conjugate must remain usable through annealing, buffer exchange, stability testing, cell exposure, and fit-for-purpose analytical confirmation.
Key Challenges Research Teams Face in Peptide–RNA Projects
Conjugation at the wrong terminus or through an overly bulky linker can interfere with hybridization, duplex formation, guide-strand behavior, or structural folding in functional RNA formats. We help evaluate where attachment should occur so the peptide improves the construct concept without disrupting the RNA role that matters most in the assay.
A cell-penetrating peptide, receptor-binding sequence, endosomal escape motif, or affinity tag can produce very different biological behavior. Projects often underperform when peptide selection is made only from popularity rather than from the intended uptake mechanism, target biology, matrix conditions, and readout strategy.
RNA is highly polyanionic, while many useful delivery peptides are cationic or amphipathic. That combination can change solubility, increase self-association, shift chromatographic retention, and make separation of free peptide, free RNA, and partially modified species more difficult than a standard oligo purification workflow.
Teams often need more than a single mass value. They need data that show conjugation state, purity, RNA integrity, residual free components, and whether the final construct is suitable for follow-on studies. We build characterization plans around the actual decision points of the project instead of treating QC as a final formality.
Our Peptide RNA Conjugation Services
We provide modular support for peptide–RNA programs ranging from first-pass feasibility assessments to conjugates prepared for expanded biological testing. Service scope can include RNA construct review, peptide and handle selection, linker planning, post-synthetic conjugation, purification development, and analytical characterization for custom research workflows.
 Construct Design Review
Construct Design Review
Capabilities include:
- Review of RNA format, strand architecture, sequence length, duplex requirements, and modification pattern before chemistry selection
- Assessment of peptide function such as cell penetration, receptor targeting, endosomal escape support, affinity capture, or encoded display
- Recommendation of 5′, 3′, or selected internal attachment strategies based on function-sensitive RNA regions
- Early risk review covering activity retention, aggregation tendency, and purification difficulty
- Comparative planning when the project may be better served by adjacent formats such as antibody-RNA conjugation or ligand-linked oligonucleotide systems
Typical applications:
Early construct screening, rescue of underperforming delivery concepts, and route planning for custom RNA research reagents
 Handle & Linker Planning
Handle & Linker Planning
Capabilities include:
- Selection of orthogonal reactive handles on the peptide and RNA to support cleaner assembly and more defined stoichiometry
- Evaluation of spacer length, hydrophilicity, and cleavable versus non-cleavable linker behavior according to the study goal
- Choice of RNA-compatible routes including activated ester approaches, maleimide conjugation, and bioorthogonal click chemistry where appropriate
- Planning around thiol state control, amine availability, steric accessibility, and metal-sensitive RNA constructs
- Route selection for direct post-synthetic assembly versus staged modular build strategies
Focus areas:
Defined attachment, preservation of RNA performance, and linker choices that remain practical for purification and follow-on assays
 Custom Peptide–RNA Assembly
Custom Peptide–RNA Assembly
Capabilities include:
- Conjugation of custom peptides to single-stranded RNA, duplex RNA components, aptamer-like RNA constructs, and other project-specific RNA formats when chemistry is feasible
- Support for CPP-linked, targeting-peptide-linked, membrane-active, affinity-tagged, or encoded-display constructs
- Development of site-selective post-synthetic assembly workflows to favor a defined product rather than broad product distributions
- Coordination of reaction conditions, solvent system, and buffer composition to reduce RNA damage and peptide-related side reactions
- Optional alignment with chemical synthesis of peptide-oligonucleotide conjugates logic when broader platform comparison is useful
Deliverables:
Custom crude or purified peptide–RNA conjugates with project-matched reaction records and recommended handling conditions
 Purification & Characterization
Purification & Characterization
Capabilities include:
- Purification planning using RP-HPLC, ion-pair or ion-exchange methods, desalting, PAGE-based cleanup, or orthogonal combinations when needed
- Analytical confirmation of conjugation state, residual free peptide or RNA, and construct purity using fit-for-purpose methods
- LC-MS, MALDI-TOF where appropriate, UV profiling, gel shift, and duplex or integrity checks selected for the actual construct type
- Comparison of candidate linker or attachment designs to identify which construct is most suitable for downstream work
- Optional support for delivery-focused studies related to siRNA delivery optimization and development when the RNA payload is used in uptake or trafficking programs
Deliverables:
Analytical summaries, purity and identity data, and recommended storage or working conditions for research use
Key Design Parameters for Peptide RNA Conjugation
Successful peptide–RNA design depends on the relationship between RNA chemistry, peptide function, attachment geometry, and the assay question the construct must answer. The table below highlights the variables that most often determine whether a conjugate is merely synthesizable or genuinely useful in downstream research.
| Design Parameter | Common Options | Development Considerations | Impact on Conjugate Performance | Why It Matters to Customers |
| RNA Format | ssRNA, siRNA strand, duplex-associated RNA, aptamer-like RNA, modified research RNA | Length, secondary structure, duplex dependency, and base or sugar modifications influence chemistry compatibility | Affects conjugation feasibility, stability, and retention of RNA function | Determines whether the proposed construct can be built without compromising the intended RNA role |
| Peptide Role | CPP, receptor-targeting peptide, endosomal escape motif, affinity tag, encoded display sequence | Charge, hydrophobicity, length, and sequence composition change both biology and manufacturability | Influences uptake behavior, target interaction, self-association risk, and purification difficulty | Helps match the conjugate to the actual research objective instead of adding a peptide without clear purpose |
| Attachment Site | 5′-terminal, 3′-terminal, selected internal handle | The chosen site must avoid critical recognition, duplexing, or folding regions | Controls how much the peptide perturbs RNA behavior and how defined the final product can be | Reduces the risk of obtaining a conjugate that is chemically valid but biologically unhelpful |
| Linker Strategy | Stable spacer, cleavable linker, hydrophilic spacer, short direct linkage | Linker length and polarity influence steric accessibility, release behavior, and chromatographic properties | Affects activity retention, construct flexibility, and assay compatibility | Often determines whether the peptide contributes useful function without masking the RNA component |
| Charge & Solubility Profile | Highly cationic peptide, amphipathic peptide, balanced neutral spacer-assisted format | Electrostatic attraction between peptide and RNA can alter aggregation and purification behavior | Influences handling, formulation window, and separation of related species | Helps avoid late-stage surprises during cleanup, storage, or cell-based testing |
| Analytical Readout Plan | LC-MS, PAGE, UV, hybridization or duplex check, orthogonal purity methods | No single method is sufficient for every conjugate architecture | Determines how confidently the intended product can be distinguished from side products | Supports project decisions with data that are relevant to the construct rather than generic release checks |
Peptide–RNA Conjugation Strategies & Process Development Considerations
No single coupling route fits every peptide–RNA project. Method selection should be guided by RNA sensitivity, peptide composition, required stoichiometry, linker behavior, purification constraints, and how the final construct will be used in downstream studies.
| Conjugation Strategy | Technical Approach | Common Applications | Development Advantages |
| Thiol–Maleimide Coupling | Cysteine- or thiol-modified peptides are coupled to maleimide-functionalized RNA or linker-bearing RNA intermediates | Defined single-site assembly, CPP-RNA constructs, modular post-synthetic build workflows | Widely used, relatively fast, and practical when a controlled 1:1 attachment route is needed |
| Azide–Alkyne Click Chemistry | Azide- and alkyne-bearing partners are joined by SPAAC or CuAAC, depending on construct tolerance and process design | Site-specific conjugates, multifunctional builds, RNA constructs requiring orthogonal chemistry | Supports modular assembly and reduces cross-reactivity with other functional groups |
| Activated Ester / Amide Linkage | Amine-modified RNA or peptide-linked carboxyl intermediates are connected through NHS-type activated esters or related linkers | Stable research conjugates, terminally modified RNA constructs, tag-bearing assemblies | Straightforward route when amine access is well controlled and orthogonal design is not overly complex |
| Disulfide-Based Attachment | Thiol-bearing partners are joined through reducible disulfide-containing linker systems | Projects evaluating triggered release or intracellularly labile attachment concepts | Useful when reversible linkage is preferable to a permanently stable construct |
| Oxime / Hydrazone-Type Ligation | Carbonyl-bearing RNA intermediates are reacted with aminooxy- or hydrazide-functionalized peptide modules | Specialized RNA conjugates, aptamer-related builds, custom research constructs with alternative handles | Expands route flexibility when thiol or click handles are not ideal for the construct |
| On-Support or Stepwise Build | Peptide and RNA elements are assembled in a staged synthesis sequence before final deprotection and cleanup | Shorter or specially designed constructs, route-development studies, difficult handle-placement problems | Can improve control over attachment position when direct solution-phase assembly is less suitable |
Analytical Characterization & Quality Control Framework for Peptide–RNA Conjugates
For peptide–RNA conjugates, analytical quality means more than showing that mass increased. The data package should help determine whether the intended product is present, sufficiently pure, structurally intact, and appropriate for the biological or mechanistic question the project is trying to answer.
| Analytical Category | Methodology | Purpose in Development | Data Delivered |
| Identity Confirmation | LC-MS, ESI-MS, or MALDI-TOF where suitable for the construct | Verifying that the expected peptide–RNA mass and conjugation state are present | Observed mass data and construct assignment summary |
| Purity Assessment | RP-HPLC, ion-pair HPLC, ion-exchange, or orthogonal chromatographic review | Separating intended conjugate from free RNA, free peptide, truncated species, and linker-related byproducts | Purity chromatograms and peak interpretation notes |
| Mobility / Integrity Check | PAGE, gel shift analysis, or other electrophoretic methods | Confirming conjugate formation and monitoring residual unconjugated components or degradation | Gel images or migration comparison data |
| RNA Structural Verification | Duplex assessment, annealing check, or project-specific hybridization readout | Showing that the RNA component remains usable after conjugation and cleanup | Duplex confirmation notes and construct handling recommendations |
| Optical / Concentration Profile | UV absorbance and, when relevant, fluorescence-based confirmation | Estimating concentration, confirming label behavior, and comparing conjugate builds | Spectral data and concentration summary |
| Stability Observation | Short-term storage checks, buffer exchange review, or stress comparisons appropriate to the project | Evaluating whether the construct remains suitable for shipping, storage, and experimental use | Stability observations and recommended handling window |
| Documentation Package | Structured reporting of build conditions, analytics, and working recommendations | Supporting repeat synthesis, transfer to internal teams, and comparison of construct variants | Conjugation summary, analytical overview, and condition recommendations |
Workflow for Custom Peptide RNA Conjugation

We begin by clarifying the RNA type, peptide role, intended assay path, and whether you already have defined handle-bearing starting materials. This step prevents chemistry development from moving ahead before the biological question and construct constraints are aligned.
Reactive handles, attachment site, spacer design, and stable versus cleavable linkage options are selected based on RNA sensitivity, peptide composition, and the level of product control the project requires.
We establish a practical assembly route and optimize reaction conditions to favor the intended conjugate while limiting RNA damage, handle hydrolysis, peptide-related side reactions, and unnecessary heterogeneity.
Free peptide, residual RNA starting material, salts, and linker-derived byproducts are removed using the purification strategy best matched to the hybrid properties of the conjugate rather than relying on a generic single-platform cleanup method.
Identity, purity, integrity, and relevant structure or duplex behavior are evaluated using fit-for-purpose analytics so candidate constructs can be compared on data that support real project decisions.
Final output may include purified peptide–RNA conjugates, analytical summaries, storage recommendations, and next-step suggestions for repeat synthesis, variant comparison, or downstream biological evaluation.
Why Choose Our Peptide–RNA Conjugation Platform
We evaluate RNA function, duplex needs, and sensitive sequence regions before defining the peptide attachment plan, which helps reduce the risk of building a conjugate that is chemically correct but functionally compromised.

We do not force all projects into one coupling route. Linker and chemistry choice are matched to the handles available, the required stoichiometry, and the tolerance of the RNA and peptide components.
Peptide–RNA conjugates often behave differently from either free peptides or free RNA. Our purification planning takes the combined charge, polarity, and aggregation tendency of the hybrid construct into account from the start.
We structure characterization around the questions that matter to the project—identity, purity, residual free components, and construct integrity—so the resulting data are easier to use in screening and follow-on planning.
Common Research Applications of Peptide–RNA Conjugates
Cellular Uptake & Trafficking Studies
- Covalent linkage of CPPs or trafficking-focused peptides to RNA constructs for uptake comparison in difficult cell models.
- Useful for studying attachment-site, linker, and peptide-sequence effects on intracellular behavior.
- Often paired with projects exploring siRNA delivery optimization and development questions.
Targeted RNA Delivery Research
- Receptor-binding peptides can be linked to RNA payloads for target-focused binding and delivery studies.
- Suitable for comparing peptide classes, linker choices, and modular construct architectures in research settings.
- Useful when defined covalent formats are preferred over loosely assembled carrier systems.
RNA Display & Selection Workflows
- Peptide-linked RNA constructs are relevant to encoded library and display formats where genotype and peptide function must remain connected.
- Conjugation control helps preserve the relationship between the RNA component and the displayed peptide sequence.
- Useful for method development, construct comparison, and workflow optimization in selection-based studies.
Aptamer & Probe Engineering
- Peptide–RNA conjugates can be used to add targeting, capture, or reporter-supporting functions to RNA probe formats.
- Applicable to custom assay reagents, binding studies, and structure-function experiments.
- Can be coordinated with broader nucleic acid labeling or related oligonucleotide modification workflows when additional tags are needed.
Discuss Your Peptide RNA Conjugation Project
Whether you are evaluating a first peptide-linked RNA concept, troubleshooting a low-yield conjugation route, or comparing multiple attachment strategies for a delivery or assay program, we provide technically focused support across design, synthesis, purification, and characterization.
Our team works with customer-defined sequences, peptide classes, linker preferences, and analytical goals to deliver conjugates and data packages that are easier to interpret and reproduce in downstream research. Contact our scientific team to discuss your peptide RNA conjugation requirements and request a project-specific proposal.
Frequently Asked Questions (FAQ)
Peptide-RNA conjugation significantly enhances RNA delivery by improving the molecule's ability to penetrate cell membranes. This process aids in more efficient cellular uptake, ensuring that RNA reaches its target cells effectively and with reduced degradation.
Peptide-RNA conjugation serves as a powerful strategy for molecular biology due to its ability to enhance RNA functionality. The process allows for RNA to be modified for better tissue penetration, stability, and controlled release, making it useful across a variety of research fields, including gene therapy, RNA vaccine development, and RNA editing.
The conjugation process boosts RNA's ability to overcome biological barriers, such as cell membranes and tissue layers, which are typically challenging for RNA-based therapies. The attached peptide acts as a facilitator, enhancing RNA's interaction with lipid-rich environments and enabling more efficient cellular uptake, allowing RNA to reach target cells, tissues, and organs that would otherwise be difficult to access.

